Get a FREE Case Evaluation
"*" indicates required fields
Quick Facts
- What is a drug? Any non-food substance that affects bodily functions and is used to diagnose, cure, mitigate, treat, or prevent a disease.
- Dangerous Drugs in the news: Depo-Provera, Suboxone, Ozempic
- Common injuries from dangerous drugs: Cancers, tumors, organ damage, vision loss, gastro-intestinal disorders, dental injuries, blood clots, cardiovascular events
- Regulator of drugs in the U.S.: U.S. Food and Drug Administration
- Medical device lawsuits filings: Lawsuits are typically filed against manufacturers who knew (or should have known) their drugs harmed people
Dangerous Drug Lawsuits
Get the Latest Updates on Dangerous Drug Lawsuits
At Lawsuit Tracker, we provide important information about drugs that have harmed people. We follow lawsuits against various drug manufacturers and share developments about those lawsuits. Our goal is to get up-to-date information to the people and families who need it most—people who have taken a potentially dangerous drug.
When we share information about a drug, that means the drug has already harmed people or may harm more people in the future. Sharing timely information is crucial to helping people understand their current or future injuries.
If you have been harmed and are considering taking legal action against the drug manufacturer who harmed you, our legal partners are ready to fight for you and change your life for the better.
Get a FREE Case Review
Contact Us NowTo help our visitors better understand how they can hold drug companies responsible for their injuries, we have created this page. The goal of sharing this information is to empower people who have been harmed by drugs that were supposed to help them.
Our Active Lawsuits
At Lawsuit Tracker, we report on dangerous drug lawsuits that matter to consumers. Below is a list of active lawsuits that our legal partners are currently taking cases on. If you have taken one of these drugs and experienced serious side effects, we want to hear from you.
Depo-Provera Lawsuit
Depo-Provera has been shown to increase the risk of a certain type of brain tumor. Many women who didn’t know their health ailments were likely caused by birth-control are now filing lawsuits.
Ozempic Lawsuit
Many people enjoy the benefits of Ozempic, but the GLP-1 drug has caused problems for some patients. People are filing lawsuits after experiencing severe gastric disorders,vision loss, and blood clots.
Suboxone Lawsuit
Suboxone films can contribute to tooth decay, leading to a range of dental injuries. Many people say they were not properly warned about how bad Suboxone strips could be for their dental health or told how they could better protect their teeth.
On this page:
Why People Sue Drug Companies
People sue pharmaceutical manufacturers when they experience severe injuries from taking a drug. When people are harmed by a drug, they file lawsuits against manufacturers for three main reasons:
- Defective design
- Manufacturing defects
- Inadequate warning labels and drug information
Defective design occurs when a drug is manufactured properly but people still experienced adverse side effects because of the contents of that drug. Manufacturing defects occur when a drug was designed properly but an error occurs during the manufacturing process. Inadequate warning occurs when a drug company did not properly warn patients about the dangers of taking a drug.
When any of these situations occur, people may be harmed by a drug that was supposed to help them. But how are people harmed by drugs that are available through their trusted doctors and pharmacists?
How People Are Harmed Despite Drug Regulations
The U.S. Food & Drug Administration (FDA) is the federal agency responsible for protecting the American public (and their pets) from dangerous drugs, foods, and medical products. The FDA has strict regulations for how drugs are approved, marketed, sold, and monitored.
Despite their efforts, some drugs are brought to market that harm people. There are many reasons for this. Drug companies often work hard to avoid disclosing side effects. Some side effects take years to present in patients (this is called a latency period). Sometimes, manufacturing plants make mistakes during the drug-making process. All of these circumstances can result in harm to patients who take these drugs.
The FDA’s Mission:
“The Food and Drug Administration is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological products, and medical devices; and by ensuring the safety of our nation’s food supply, cosmetics, and products that emit radiation.”
Source: https://www.fda.gov/about-fda/what-we-do

The FDA’s Role in Protecting People from Dangerous Drugs
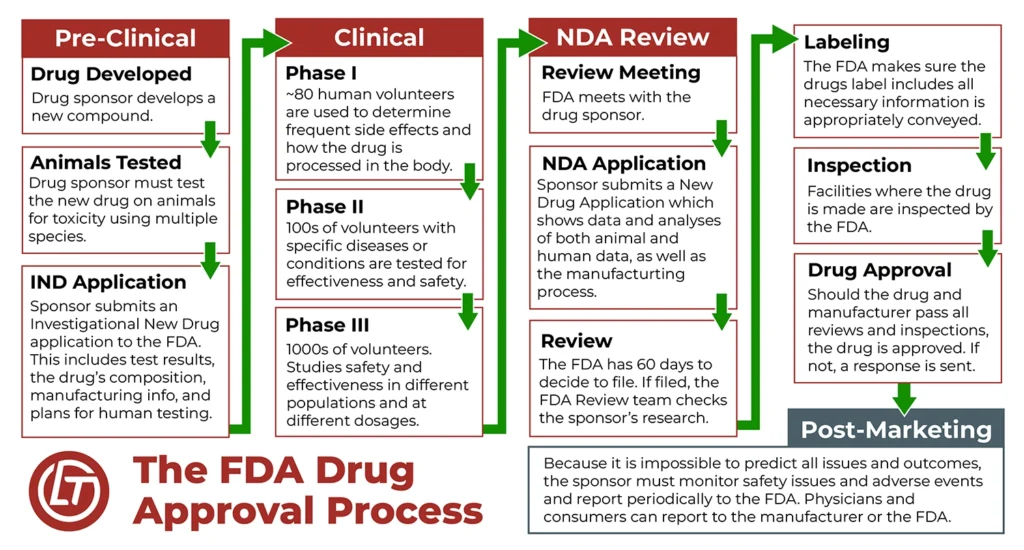
In order to be prescribed to patients, drugs have to go through a rigorous FDA approval process. But that approval process is just the first step in protecting people from harmful drugs and side effects. The FDA also enforces drug safety by:
- Conducting clinical trials
- Conducting post-approval monitoring of patients
- Inspecting manufacturing facilities to enforce FDA standards
- Issuing drug recalls when safety is in question
- Informing the public about emerging drug-safety concerns (e.g., safety alerts)
- Requiring manufacturers to update drug warning labels
- Removing dangerous drugs from the market
Even with these safety measures in place, people still experience injuries and serious side effects from taking pharmaceutical drugs. As people take medications and experience side effects, the FDA has other monitoring mechanisms in place. Two of those are an adverse-event-reporting system and on-going research from the scientific community.
MedWatch and the FDA Adverse Event Reporting System (FAERS)
The FDA maintains a public database that houses complaints filed by doctors and the general public. The system is known as FAERS, and it is part of the FDA’s post-marketing safety surveillance program. Patients and doctors can report concerns and issues through the MedWatch reporting program.
If someone experiences an adverse event (injury) after taking a drug or using a therapeutic biologic product, they can report it to the government. The FDA monitors for similar events that may be cause for concern. When the FDA gets numerous complaints about the same drug, they often launch an investigation. Drug manufacturers also receive the complaints filed in the database, to warn them of potential serious developments.
If you experienced serious side effects from a drug, you can report them to the FDA.
Medical Studies
The medical and research communities are very active in monitoring adverse drug reactions (ADRs) after a drug is on the market. Scientists and doctors study things like drug effectiveness, positive outcomes, and negative outcomes. Scientists from around the globe conduct research and publish that information in reputable journals and other scientific publications.
The FDA relies on that research to make initial drug approvals and make changes after a drug is available. Sometimes important research is published that leads to the FDA taking action against a drug manufacturer or removing a drug from the market.
Read more about how the FDA uses scientific research in its approval and surveillance processes.
Are Drug Companies Required to Protect Consumers?
The FDA requires that all pharmaceutical manufacturers adhere to specific regulations. In order for a drug to be FDA-approved, a drug company must prove its drug is safe and effective. Drug companies also have to prove they can manufacture their drugs according to federal quality standards. Once a medicine is approved, drug companies have additional responsibilities.
The Current Good Manufacturing Process (CGMP)
All drug manufacturers must adhere to the FDA’s CGMP. This process contains the minimum actions drug companies must take when creating, manufacturing, processing, and packaging their products.
Drug Company’s Rules for Marketing and Advertising
Drug companies have to follow strict rules when advertising and marketing their drugs. The FDA sets specific standards to make sure all drug marketing follows federal codes and regulations. However, drug companies can still market drugs, even ones with serious adverse reactions and side effects.
Does Federal law ban ads for drugs that have serious risks?
“No. Federal law does not bar drug companies from advertising any kind of prescription drugs, even ones that can cause severe injury, addiction, or withdrawal effects.”
Source: FDA’s prescription drug advertising questions and answers
Responding to Reports of Adverse Drug Events
When the FDA receives reports of adverse reactions by way of its MedWatch Program, the drug manufacturer has to take action. Depending on how often and how serious the events are, drug manufacturers are legally required to act. These required actions could include:
- Conducting scientific investigations
- Updating a drug’s warning label
- Issuing a safety alert
- Removing a drug from the market
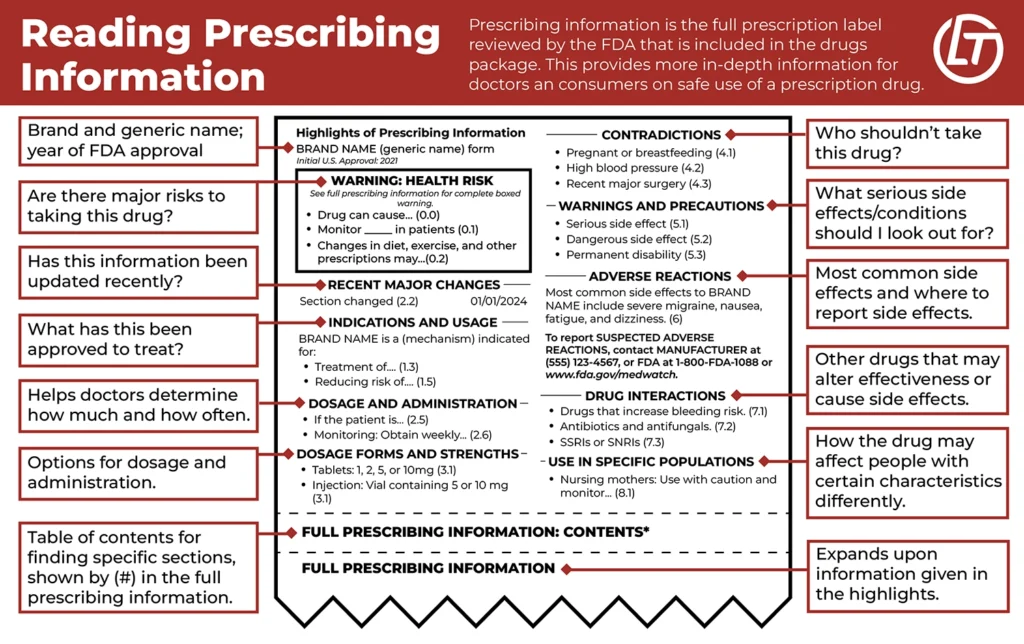
Drug Labels, Inserts, and Directions
When a patient takes or a doctor administers a medication, it is the responsibility of the drug manufacturer to supply information about potential side effects, contraindications, and prescribing information. By supplying this information, patients and doctors can make informed decisions about whether or not taking a drug is in the best interests of the patient. However, drug companies sometimes go to great lengths to keep important information out of the hands of patients and doctors. Other times, they are not aware of the specific danger a drug presents. When patients aren’t properly warned and informed, drug companies can be held liable.
Why Drug Companies Can Be At Fault for Harming People
Drug companies have certain duties, as determined by the FDA. When they do not uphold those duties, people get sick or are hurt by medications that are supposed to help them. When that happens, drug companies can be determined to be at fault for their negligence. There are many reasons a pharmaceutical company can be held legally liable.
For each drug a company makes, they are required to exercise reasonable care in the following ways:
- Designing
- Formulating
- Manufacturing, storing, distributing
- Compounding
- Testing
- Inspecting
- Labeling
- Packaging
- Distribution
- Marketing, promotion, advertising, and sales
- Research and testing of post-approval safety
If a pharmaceutical company fails to uphold any of these duties, they could be held liable for their failures. Drug companies are presumed to be experts in their field.
Through lawsuits, it can be determined that drug companies either were aware of the dangers of their drugs or should have known about the dangers of their drugs. For example, a company should be following developments from the scientific community. If a company receives information from the FDA’s reporting systems, the independent scientific community, or their own researchers, they need to take action. If they fail to take action, they may be found liable for the injuries their drugs cause.
What Are the Reasons People Sue Drug Companies?
When drug manufacturers do not hold up their responsibilities, innocent people are harmed. When people experience serious, life-changing injuries because of a drug company’s failures, they often have to pursue legal action. When injured people file lawsuits, they become plaintiffs. Here are some the ways drug companies are accused of being at fault for a plaintiff’s injuries:
- Failure to Warn
- Negligent Failure to Warn
- Design Defect
- Negligence
- Negligent Design Defect
- Negligent Misrepresentation
- Fraudulent Misrepresentation
- Breach of Express Warranty
- Breach of Implied Warranty
All of these accusations imply that a drug company should have done something to protect the consumer, but failed to do so.
Types of Lawsuits People File Against Drug Companies
When someone is harmed or killed by a drug or medical product, that person or their surviving family members may file a lawsuit against a drug company. Depending on the circumstances surrounding the injury and timeline of the lawsuit, there are different types of legal actions people can take. Those actions include filing a personal injury lawsuit, filing as part of a mass tort, or filing as part of a class action lawsuit.
Drug Companies and Personal Injury Lawsuits
When people experience serious injuries after taking a drug, they may choose to file a civil lawsuit against that drug’s manufacturer. This is more likely to occur if the lawsuit has not been consolidated into a multidistrict litigation or class action. This type of lawsuit is often referred to as a product liability lawsuit. In this type of lawsuit, a single injured party (the plaintiff) takes a solo legal action against a drug company (the defendant). Depending on the outcome, the plaintiff may receive compensation for their injuries.
Drug Companies and Mass Torts
When a drug harms many people in the same way, those cases may become part of a mass tort. Mass torts occur when multiple people are harmed by the same action, event, or omission made by a drug company. Mass torts may list one or more drug companies as defendants.
The goal of a mass tort is to combine multiple, similar court cases into a single legal proceeding. This can speed up the lawsuit process for the plaintiff, defendant, and the courts.
In a mass tort proceeding, each person who is harmed (plaintiff) is treated as an individual. Their case is tried individually and their unique circumstances and injuries are reviewed. However, many of the early steps of the legal process have already been established, saving everyone involved time and effort.
Assuming the plaintiff’s lawyer can prove their client’s injuries were caused by a drug, they can receive a settlement. Some mass tort cases do go to trial.
Mass Tort Fact:
Successful plaintiffs receive individual settlements or awards based on their unique circumstances and injuries.
Drug Companies and Multidistrict Litigation
When people who are harmed by the same drug in a similar way, their cases often become part of a Multidistrict Litigation (MDL). An MDL is a type of federal civil lawsuit that is consolidated into a single, federal court. This streamlines the legal process for all parties.
MDL Fact:
Unlike a class action lawsuit, successful plaintiffs in an MDL receive compensation based on the extent of their individual injuries. That money often comes from a settlement, but settlements vary from person to person.
Drug Companies and Class Action Lawsuits
Class action lawsuits against drug manufacturers do happen, but they are not as common for people who are harmed after taking a drug. In a class action lawsuit, the lead attorneys represent the entire group of plaintiffs. All plaintiffs share the same evidence. Their cases are not litigated individually. Depending on the case, the plaintiffs may have to join the class action, or they may be automatically added to it.
Class Action Fact:
Each plaintiff in a class action lawsuit typically receives the same amount of money. Their individual circumstances are not considered by the courts.
How Our Legal Partners Can Help People Harmed by Pharmaceutical Drugs
If you or a family member experienced serious side effects after taking a prescription drug, our team may be able to help you recover money. At Lawsuit Tracker, we are a team of legal partners who represent people harmed by dangerous or defective drugs.
Our lawyers can help you and your family get back on their feet. Our job is to prove how your injuries were caused by a drug company’s negligence and failures. Our legal partners have years of experience in mass torts, and we take our jobs very seriously.
Attorneys for People Harmed by Dangerous Pharmaceutical Drugs
If you have been harmed by a prescription drug and are interested in learning more about your legal options, contact our team. We can work with you to determine if you qualify to file a claim. If you decide you are ready, we are ready to fight for you and help you get the compensation you deserve.
Get a FREE Case Review
Contact Us TodayWhat People Harmed By Drugs Can Receive Compensation For
When drugs harm people, their entire lives are affected. The financial and personal losses can be staggering, and our team is here to help offset those losses. If you choose to file a lawsuit against a drug company, you may receive compensation related to the following damages:
- Medical expenses (e.g., hospital stays, treatments, doctor visits, medical devices, rehabilitation)
- Lost wages
- Pain and suffering
- Emotional distress
- Future medical costs
- Loss of consortium (negative impacts to your relationship with your spouse)
Our team understands the serious impacts these losses have on your life. We are ready to fight for you.
Contact Us Today
"*" indicates required fields