Get a FREE Case Evaluation
"*" indicates required fields
Quick Facts
- Brand Names: Physiomesh, C-QUR mesh, Composix Kugel patch, and others
- Device Used For: Hernia repair
- Product type: Surgical mesh
- Manufacturers With Lawsuits: C. R. Bard, Covidien, Atrium, Ethicon
- Potential Severe Complications: Infections, product migration, adhesion to organs, product shrinkage or breakdown
- Lawsuit Type: Multidistrict litigation
- Lawsuit Status: Active
Hernia Mesh Lawsuit – April 2025 Update
Individuals are filing hernia mesh lawsuits against multiple manufacturers, including Bard, Covidien, Ethicon, Atrium Medical Corporation, Gore Medical, and Genzyme. These lawsuits are being filed after people experienced infection, migration, rejection, bowel obstruction, and other serious complications from hernia mesh. These lawsuits have been consolidated into four multidistrict litigations (MDL# 2782 in the Northern District of Georgia, MDL# 2846 in the Southern District of Ohio, MDL# 2753 in the District of New Hampshire, and MDL# 3029 in the District of Massachusetts.)
As of November 2024, there are over 26,000 lawsuits pending in the federal hernia mesh litigation. The presiding judge is the Honorable Edmund A. Sargus in the largest MDL against C.R. Bard. This lawsuit is in the late stages, and an undisclosed settlement has been reached to resolve most claims. Other lawsuits, however, are in the mid-stages, with bellwether trials expected to happen by summer 2025. People are continuing to file hernia mesh lawsuits today. Due to the impending resolution of these claims, immediate action is critical.
Hernia Mesh Lawsuit Timeline & News – April 2025
March 12, 2025: Progress Made on Bard Hernia Mesh Settlements
People harmed by Bard hernia mesh products and their lawyers are looking at their options for pursuing a settlement. Qualified patients have three settlement options: quick-pay 1, quick-pay 2, or traditional pay. The quick-pay 1 option is for people implanted with a hernia mesh covered by the settlement who do not have a qualifying injury. Quick-pay 2 is for people with hernia mesh and basic injuries covered by the settlement. Finally, traditional pay will be for the most serious hernia mesh injury cases. Patients and their attorneys will be examining the specifics of each case and determining the best path forward. Some people may still be able to file a lawsuit against a hernia mesh manufacturer and pursue their own settlement options.
March 11, 2025: Expert Evidence Due In Covidien Hernia Mesh Lawsuits
Multiple facets of the Covidien hernia mesh litigation is moving forward. Expert deadlines are here. We are eager to hear whether there might still be a bellwether trial this year. Bellwether trials are test trials that often predict future success for many plaintiffs in the litigation. The parties (i.e., lead lawyers for the plaintiffs and Covidien’s lawyers) also agreed on an experienced mediator. A mediator is a positive development for a potential settlement. Covidien is defending on three fronts, Minnesota state court, Massachusetts state court, and the federal lawsuits pending in the District of Massachusetts.
March 6, 2025: Hernia Mesh Claims Against C.R. Bard and Davol Increase as Settlements Are Underway
The U.S. Judicial Panel on Multidistrict Litigation allowed five more cases to join the federal MDL against hernia mesh manufacturers like C.R. Bard and Davol. Patients and their surviving loved ones are speaking out after hernia mesh products caused them severe injuries. Some hernia mesh can adhere to or cut through organs or tissue or migrate to other spots, which can be dangerous and, in some cases, life-threatening. Bard reached a settlement to resolve many of the claims currently pending against it; however, more plaintiffs can file lawsuits if they were injured by hernia mesh made by Bard, Davol, or another pharmaceutical company.
February 25, 2025: Settlement Deadline in Hernia Mesh Lawsuits Extended
The Bard hernia mesh settlements have experienced some delays. The crosslink portal that plaintiffs must use to upload medical records and documents to prove they qualify for the settlement has been having some issues. The deadline to file for the Bard hernia mesh settlement was supposed to be March 17, 2025, but the date has been pushed back several months because of technical problems. The indications remain that settlement money will be awarded on a first-come-first-serve basis, so plaintiff lawyers are still doing their best to submit proof of 1) defective bard hernia mesh 2) injury caused by the defective hernia mesh.
February 10, 2025: C.R. Bard likely to Settle Some Current Hernia Mesh Lawsuits
Currently, 24,102 hernia mesh lawsuits are pending against C.R. Bard. After the January 2025 settlement order, it is likely some of these lawsuits will reach settlement. Hernia mesh lawsuits are still being actively filed against different manufacturers of these products. Covidien has 1,660 lawsuits pending, Atrium has 500 cases, and there are 16 left for the Ethicon lawsuit that has already settled thousands of cases for the injuries caused by their mesh.
February 7, 2025: More Consumers File Hernia Mesh Lawsuits After Bard Reaches Settlement
C.R. Bard reached a settlement agreement last month, which may resolve many of the 24,102 cases against it for its defective hernia mesh products. Ethicon also settled or resolved a large number of the 4,000 claims that have been filed against it since the litigation began. Other hernia mesh manufacturers are managing thousands of allegations lodged by consumers or their surviving loved ones. Covidien has 1,660 lawsuits against it, and Atrium has 500 that are pending. More plaintiffs can join in this lawsuit if they were harmed or their loved one was killed by defective hernia mesh.
January 7, 2025: Intensive Settlement Process Begins For Hernia Mesh Settlements
In December of 2024, progress was made for people who are pursuing settlements from Bard. Case Management Order #55 appointed special masters and implemented an Intensive Settlement Process (ISP) to begin resolving hernia mesh cases. A settlement portal was also opened for people who want to begin filing for settlements for injuries incurred from their hernia mesh implants. Special Masters Ellen K. Reisman and John Jackson will oversee the process. The ISP will not begin until January of 2027. At that point, people who want to opt out of a trial and pursue a settlement can begin attending settlement hearings and negotiations. Although the timeline is likely frustrating for many people who suffered hernia mesh injuries, this ISP is a step closer to compensation and closure.
November 5, 2024: 10 Lawsuits Dismissed from Ethicon MDL After Settlement
An additional 10 cases have been dismissed from MDL No. 2782 after agreeing to a confidential settlement. The lawsuits filed against Johnson & Johnson subsidiary Ethicon were related to the company’s physiomesh flexible composite hernia mesh products.
October 20, 2024: Thousands of Hernia Mesh Cases Expected to be Filed in Coming Years
Despite settlements and a growing trend for confidential agreements to resolve lawsuits behind closed doors, hernia mesh lawsuits remain active. Legal experts expect thousands of more cases to be filed in the next several years as many potentially defective products remain on the market.
October 15, 2024: Wife Takes Action in Bard Hernia Mesh MDL on Spouse’s Behalf After His Death
The wife of a plaintiff in the Bard Hernia Mesh has taken action after his untimely death. According to a court order filed on October 15, 2024, Joyce Mabry, the spouse of William Mabry, will stand in the place of her husband in the lawsuit filed against C.R. Bard. Mrs. Mabry has also filed a wrongful death claim on behalf of her husband.
October 2, 2024: Settlement Reached in MDL No. 2846
A long-awaited settlement has been reached in MDL No. 2846 against C.R. Bard. Parent company Becton, Dickinson and Company announced via a press release that it had reached an agreement to resolve most of the pending hernia mesh lawsuits. Exact payouts remain uncertain, but more details are expected to emerge over the coming weeks and months.
October 1, 2024: Qualified Settlement Fund Approved in Atrium Hernia Mesh MDL
Judge Landya B. McCafferty approved a qualified settlement fund in MDL No. 2753 against Atrium Medical Corporation for their C-Qur Mesh products. The confidential settlement agreement will resolve some of the more than 600 claims in the litigation.
On this page:
Hernia Mesh Lawsuit Timeline & News – April 2025
What is the Hernia Mesh Lawsuit About?
Why Are People Filing Hernia Mesh Lawsuits?
Whom Are People Filing Hernia Mesh Lawsuits Against?
What Stage is the Hernia Mesh Lawsuit In?
What Is Hernia Mesh and How Does It Work?
Injuries Caused By Hernia Mesh
Studies Linking Hernia Mesh and Intestinal Complications
Current/Past Hernia Mesh Information Labels
FDA/EPA Warnings About Hernia Mesh
What Damages Can People Sue Hernia Mesh Manufacturers For?
Who Qualifies to File a Hernia Mesh Lawsuit?
7 Steps to File a Hernia Mesh Lawsuit
What is the Deadline for Filing a Hernia Mesh Lawsuit?
What is the Hernia Mesh Lawsuit About?
Thousands of individuals nationwide have filed lawsuits against the manufacturers of hernia mesh products that have caused harm after implantation. The injuries associated with these products include infection, migration, adhesions, organ perforation, and hernia recurrence. In many cases, the injuries required revision surgery. In their lawsuits, injured patients allege the manufacturers of these products knew or should have known of their dangers and failed to warn consumers.
| Hernia Mesh Lawsuit Information | |
| Lawsuit Name: | IN RE: Davol, Inc./C.R. Bard, Inc., Polypropylene Hernia Mesh Products Liability Litigation |
| Main Injuries: | Adhesion, infection, mesh migration, bowel obstruction, organ perforation, bleeding, and rejection |
| Defendants: | Bard (formerly known as C.R. Bard, owned by Becton Dickinson) |
| Mass tort or class action? | Mass Tort |
| MDL Number: | 2846 |
| Pending Cases: | 24,099 |
| Court Name (Venue): | Southern District of Ohio |
| Have There Been Settlements? | Yes |
| Active Lawsuit? | Yes |
| Hernia Mesh Lawsuit Information | |
| Lawsuit Name: | In Re: Covidien Hernia Mesh Products Liability Litigation No. II |
| Main Injuries: | Adhesion, infection, mesh migration, bowel obstruction, organ perforation, bleeding, and rejection |
| Defendants: | Covidien owned by Medtronic |
| Mass tort or class action? | Mass Tort |
| MDL Number: | 3029 |
| Pending Cases: | 1,546 |
| Court Name (Venue): | District of Massachusetts |
| Have There Been Settlements? | No |
| Active Lawsuit? | Yes |
| Hernia Mesh Lawsuit Information | |
| Lawsuit Name: | In Re: Atrium Medical Corp. C-Qur Mesh Products Liability Litigation |
| Main Injuries: | Adhesion, infection, mesh migration, bowel obstruction, organ perforation, bleeding, and rejection |
| Defendants: | Atrium Medical Corporation |
| Mass tort or class action? | Mass Tort |
| MDL Number: | 2753 |
| Pending Cases: | 620 |
| Court Name (Venue): | District of New Hampshire |
| Have There Been Settlements? | Yes |
| Active Lawsuit? | Yes |
| Hernia Mesh Lawsuit Information | |
| Lawsuit Name: | In RE: Ethicon Physiomesh Flexible Composite Hernia Mesh Products Liability Litigation |
| Main Injuries: | Adhesion, infection, mesh migration, bowel obstruction, organ perforation, bleeding, and rejection |
| Defendants: | Ethicon (a Johnson & Johnson subsidiary) |
| Mass tort or class action? | Mass Tort |
| MDL Number: | 2782 |
| Pending Cases: | 26 |
| Court Name (Venue): | Southern District of Georgia |
| Have There Been Settlements? | Yes |
| Active Lawsuit? | Yes |

Why Are People Filing Hernia Mesh Lawsuits?
Tens of thousands of individuals across the country have filed lawsuits against the manufacturers of hernia mesh, alleging hernia mesh products are defective and cause harm. Legal filings indicate that plaintiffs suffered severe harm, including organ perforation, infection, mesh migration, and adhesion, as a result of the flawed design. In some instances, patients have had to undergo surgery to repair the damage caused by the medical device. Tragically, there have been deaths that are believed to be related to some of the now-recalled hernia mesh products.
Accusations against the manufacturers include that they knew or should have known about the risks of these mesh devices and failed to warn consumers. Plaintiffs also allege the products themselves were defectively designed. In addition to the four multidistrict litigations filed across the country with more than 26,000 pending cases, claims have been filed in state courts. The litigation remains active and ongoing. Affected individuals may be eligible for compensation to cover any medical expenses, lost wages, wrongful death claims, and more.
Hernia Mesh Lawsuit Spotlight
Stinson v. Davol, Inc., et al. (18-cv-1022)
On October 16, 2023, jury trial started in the Stinson v. Davol, Inc., et al. (18-cv-1022) case. The matter marked the third bellwether trial in the Bard MDL. The plaintiff, Aaron Stinson, had a Bard PerFix device implanted to repair an inguinal hernia in 2015. After the surgery, Stinson had chronic pain, which was later determined to be caused by the mesh implant. The device had deformed, curling into a ball and creating scar tissue. Despite surgery to remove the defective mesh, Stinson’s pain remained. On November 8, 2023, a jury found in favor of Mr. Stinson, awarding him $500,000 for his losses.
Whom Are People Filing Hernia Mesh Lawsuits Against?
Individuals who have suffered harm after having hernia mesh implanted are filing lawsuits against the companies that manufactured and sold the products. The hernia mesh lawsuits are unique because of the large number of defendants who manufactured and sold potentially defective products. Several of the defendants are subsidiaries of or wholly owned by pharmaceutical and medical technology giants, such as Becton Dickinson and Johnson & Johnson.
These are the defendants in the hernia mesh lawsuit:
- Bard (formerly known as C.R. Bard, now owned by Becton Dickinson)
- Covidien (owned by Medtronic)
- Ethicon (a Johnson & Johnson subsidiary)
- Atrium Medical Corporation
- Gore Medical (part of W. L. Gore & Associates, Inc)
- Genzyme (owned by Sanofi)
Information for some of the defendants is listed below.
Defendant: Bard
People harmed by hernia mesh are seeking damages against Bard in MDL 2846, Davol, Inc./C.R. Bard, Inc., Polypropylene Hernia Mesh Products Liability Litigation.
C.R. Bard, Inc. is the developer, manufacturer, and marketer of medical technology. In December 2017, Becton, Dickinson and Company acquired Bard. Prior to the acquisition, Bard manufactured and sold Polypropylene Hernia Mesh Products. These products were implanted in thousands of patients around the country due to reports of serious adverse reactions, including bowel perforation, mesh migration, and adhesion.
In 2006, several Bard hernia mesh products were the product of a Class 1 recall. Despite concerns over the safety of their medical devices, Bard continued to develop, manufacture, and market hernia mesh products, many of which remain on the market today.
Defendant: Covidien
People harmed by hernia mesh are seeking damages against Covidien in MDL 3029, IN RE: Covidien Hernia Mesh Products Liability Litigation (No. II).
Covidien, a global healthcare company headquartered in Ireland, was purchased by Medtronic in 2015. Before the acquisition, Covidien manufactured and sold hernia mesh products that are now the subject of pending litigation in the District of Massachusetts. It is alleged that the hernia mesh produced by Covidien caused serious harm to patients. The products in question are the Parietex and Parietene mesh models, which are prone to deterioration, according to court documents.
Defendant: Atrium
People harmed by hernia mesh are seeking damages against Atrium in MDL 2753, IN RE: Atrium Medical Corp. C-Qur Mesh Products Liability Litigation.
Atrium Medical Corporation is the manufacturer of C-Qur mesh, a polypropylene hernia mesh recalled by the FDA in 2013. The recall occurred after it was determined that the mesh would adhere to the package liner when exposed to high humidity. Atrium has settled some of the lawsuits that were consolidated into the MDL in the District of New Hampshire, but not all cases have been resolved.
Defendant: Ethicon
People harmed by hernia mesh are seeking damages against Atrium in MDL 2782, IN RE: Ethicon Physiomesh Flexible Composite Hernia Mesh Products Liability Litigation.
Ethicon, Inc. is a subsidiary of pharmaceutical giant Johnson & Johnson. Ethicon is the manufacturer of Physiomesh Flexible Composite Hernia Mesh. The mesh is at the center of over two dozen active cases that have been consolidated in multidistrict litigation out of the Northern District of Georgia. The company is coming under scrutiny for allegedly failing to inform consumers about the risks associated with its product.
Allegations Against These Manufacturers
Allegations against hernia mesh manufacturers, including Bard, Atrium, Ethicon, and Covidien, include that the product contained a design defect that caused direct harm to patients. In some instances, patients had to have revision surgery to repair or remove the damaged mesh. Additionally, plaintiffs allege that the companies were negligent and that their negligence resulted in actual damages to patients. Legal documents accuse the corporations of failing to warn consumers about the potential dangers associated with the use of the mesh.
What Stage is the Hernia Mesh Lawsuit In?
Hernia mesh lawsuits are in the mid-to-late stages, depending on the jurisdiction. In October 2024, the parent company of Bard announced a settlement agreement had been reached in the largest hernia mesh MDL in the country. While details of the settlement agreement amounts remain scarce, estimated payouts are expected to range between $50,000 and $100,000 depending on the severity of the injuries. Due to the complexities of a hernia mesh lawsuit and the stage of the litigation, it is strongly recommended that any aggrieved patient speak to an attorney immediately.
What Is Hernia Mesh and How Does It Work?
Hernia mesh is a type of surgical mesh used to repair hernias or an area in the body where the internal organ or fatty tissue has pushed through a weakened part of the muscle. One of the most common ways to repair a hernia is with the assistance of surgical mesh. Hernia mesh is generally made using synthetic materials to reinforce the weakened area.
Surgical mesh is used to repair hernias, however, can cause serious problems. The materials may adhere to the organs, causing perforation. Depending on the type of mesh, it may be prone to migration or deterioration, causing infection and other adverse reactions.
What is Hernia Mesh Made Of?
Hernia mesh is generally made of synthetic materials. In some cases, it may be made of animal tissue. The current lawsuits surrounding hernia mesh products involve devices made from different materials. Bard’s hernia mesh devices were made using polypropylene, a material that plaintiffs argue is unsuitable for implantation and degrades over time. Other materials are subject to shrinkage or migration.
Why Did Some Hernia Mesh Products Fail?
It is alleged that the materials used in some of the now-recalled hernia mesh products were prone to failure either due to degradation or unsuitability. Plastics used in some of the products may cause infection in the body, whereas other meshes adhere to surrounding organs, resulting in perforation. Patients have also reported shrinkage and deformation of the mesh after surgery, resulting in debilitating pain.
Hernia Mesh Complications
Hernia mesh comes with significant complications, which may require revision surgery for repair. Lawsuits filed against the manufacturers of hernia mesh allege that the companies knew or should have known of the risks of using the device but failed to warn consumers.
Bard polypropylene mesh may cause inflammation, recurrence of the hernia, adhesions, erosion, and migration of the mesh. It is alleged, through court documents, that the material used in these devices may make them more prone to adverse reactions. Covidien also manufactured hernia mesh made of polypropylene plastic, which is known to deteriorate.
Ethicon Physiomesh Flexible Composite Mesh was shown to have a higher rate of recurrence or reoperation compared to similar products. It is believed that the materials used in the mesh make it more likely to degrade, requiring revision.
Lawsuits filed against Atrium Medical Corporation allege that the use of Omega-3 fish oil to coat the C-Qur hernia mesh can cause serious complications because it adheres to the package lining when exposed to humidity.
Long-term Complications of Hernia Mesh:
- Infection
- Migration
- Adhesion
- Bowel or organ perforation
- Bleeding
- Shrinkage
- Rejection of the mesh
- Chronic pain
- Hernia recurrence
- Fistulae
- Erosion
Common Complications of Hernia Mesh:
- Temporary pain
- Fluid build-up
- Discomfort
- Scar tissue
- Irritation of the bowels
Serious Side Effects Associated with Hernia Mesh Lawsuit
Serious side effects associated with the hernia mesh lawsuit include bowel perforation or migration of the mesh. Any mesh failure, such as adhesion to the wrong part of the body or degradation of the material itself, can cause severe complications that may require surgical intervention to repair.
Injuries Caused By Hernia Mesh
Lawsuits filed against the manufacturers of hernia mesh devices allege that the products caused severe and, in some cases, irreversible harm. While rare, some injuries require surgery to correct or repair the condition. For patients with serious side effects, their lives are often deeply impacted due to chronic pain and discomfort.
Pain, Discomfort or Tenderness
When hernia mesh fails, it may cause pain or discomfort. While some degree of tenderness is to be expected after any surgery, victims of hernia mesh failure often experience chronic, debilitating pain that can only be corrected through the removal of the mesh and revision surgery.
Outcomes for this injury
Long-term or severe pain is not a normal side effect of hernia mesh surgery and should be reported to your healthcare provider. Depending on the severity of the issue, the mesh may have to be removed and revision surgery completed.
Flu-like Symptoms
Flu-like symptoms following a hernia repair are generally a sign of infection. Concerning symptoms include fever, fatigue, body aches, or swollen lymph nodes. Infections caused by hernia mesh, while rare, can become serious and potentially life-threatening.
Outcomes for Flu-like Symptoms
Any new or worsening symptoms could be a sign of infection and should be treated immediately. The prognosis for an infection caused by hernia mesh is generally good as long as it is caught early.
Nausea and Vomiting
Nausea and vomiting following the implantation of hernia mesh may be an indication of infection or device failure. Symptoms should be monitored closely and reported to a healthcare provider.
Outcomes for Nausea and Vomiting
While nausea and vomiting are not considered life-threatening in most cases, it may be a sign of a more serious problem. It is believed that in many instances, the materials used to make the mesh may be unsuitable for implantation in humans and can cause infection, fluid build-up, and other issues.
Swelling, Redness or Heat Near Implant
Swelling, redness, or heat near the hernia mesh implant is likely an indication of infection or inflammation. Prompt diagnosis and treatment are often critical for a successful outcome.
Outcomes for Swelling, Redness, or Heat Near Implant
Chronic infection and inflammation are a potential side effect of hernia mesh implantations. While the outcome is generally good with prompt treatment, patients may experience a diminished quality of life due to debilitating pain and discomfort.
Lethargy, Depression, and Sleep Issues
Long-term lethargy, depression, or sleep issues after surgery may be related to mesh failure. Litigation currently pending against multiple hernia mesh manufacturers suggests that the medical devices may be prone to failure, including adhesion, migration, and deterioration.
Outcomes for Lethargy, Depression, and Sleep Issues
In many instances, symptoms of lethargy, depression, or sleep disturbances can be corrected through the removal of the failed device or revision surgery. Unfortunately, several of the devices are known to have high rates of recurrence or repair.
Erectile Dysfunction
Patients, particularly those with inguinal hernias, may suffer erectile dysfunction as a result of irritation caused by the mesh. A study published in the Journal of the American College of Surgeons found that sexual dysfunction and pain with sexual activity were not rare after inguinal hernia repair. Over 5% of men who went through the operation experienced sexual dysfunction, which may be a result of inflammation caused by the mesh.
Outcomes for Erectile Dysfunction
Erectile dysfunction is a common side effect of inguinal hernia repair. While additional studies are needed to determine what causes the dysfunction, men should report any abnormalities, pain, or discomfort to their healthcare provider. Some sexual dysfunction or pain with sexual activity may be temporary, while others may require surgery to repair or replace failed mesh.
Bowel Obstructions and Constipation
Hernia mesh that is improperly placed or subject to migration, degradation, erosion, or adhesion may cause a bowel obstruction. Symptoms may be constipation or the inability to have a bowel movement. Small bowel obstruction is more common in laparoscopic inguinal hernia repair.
Outcomes for Bowel Obstructions and Constipation
Bowel obstruction caused by misplaced or defective hernia mesh can range in severity. Depending on the obstruction, a doctor may need to remove the mesh and potentially some of the damaged portion of the bowel. Removal of the bowel can significantly impact a person’s quality of life and may inhibit them from performing their normal daily activities.
Recurrence of Hernia
While the rate of recurrence varies, any failure of the mesh could result in the need for a secondary surgery to repair or replace the device. It is alleged that manufacturers knew or should have known that their products contained a design defect, which made them more prone to failure and caused them to fail to warn consumers. This resulted in a higher rate of recurrence.
Outcomes for Recurrence of Hernia
While the outcomes for recurrence of hernia are generally good, they may require revision surgery to replace or repair the issue. Surgery often creates scar tissue, which can cause pain and discomfort, even years after the initial procedure.
The FDA wants to receive reports of injuries and side effects people experience after using a medical device. The FDA Adverse Event Reporting System (FAERS) informs future decisions about drugs. If you have a hernia mesh injury you want to report, you can learn more about submitting a Medwatch Voluntary Report here.
Signs and Symptoms of Hernia Mesh Failure
In the weeks and months following a hernia repair, it is imperative to watch for new and worsening symptoms, which may be a sign of mesh failure. Mesh failure may present itself in a number of ways, including through inflammation, flu-like symptoms, pain, and discomfort. Prompt diagnosis and treatment of any hernia mesh-related injuries are often critical for a successful outcome.
Signs and symptoms of hernia mesh failure:
- Hernia recurrence
- Signs of infection, i.e., flu-like symptoms
- Nausea and vomiting
- Pain or discomfort
- Tenderness at the place of implantation
- Swelling or redness near the implant
- Warmth near the surgical mesh
- Depression
- Lethargy or trouble sleeping
- Constipation
- Bowel obstruction
- Erectile Dysfunction
Studies Linking Hernia Mesh and Intestinal Complications
Research indicates that hernia mesh, particularly mesh made from certain plastics such as polytetrafluoroethylene (PTFE)/polypropylene, may have an increased risk of intestinal complications. Patients who had surgical mesh implanted during hernia repair were also at a higher risk of infection.
Important Study: Major mesh-related complications following hernia repair: events reported to the Food and Drug AdministrationIn December 2005, researchers published findings regarding the increased risk of significant mesh-related complications following hernia repair in the peer-reviewed journal Surgical Endoscopy. A review of over 250 adverse event reports filed with the U.S. Food and Drug Administration found that 42% of people filing a report suffered infection, 18% had mechanical failure, 7% experienced intestinal complications, and 6% suffered from adhesions.
| Overview: Major mesh-related complications following hernia repair: events reported to the Food and Drug Administration | |
| Description of Study: | A review of the adverse event reports filed with the FDA regarding hernia mesh complications. |
| Published In: | Surgical Endoscopy |
| Study Authors | T N Robinson, J H Clarke, J Schoen, and M D Walsh |
| # of Participants | 252 |
| Findings | The study found that infection and mesh failure were the most-common injuries reported. |
Hernia Mesh Recalls
Over 200,000 hernia mesh units have been the subject of recalls over the past 20 years. Recalls were initiated for a number of reasons, including mislabeling, counterfeit lots, and an increased risk for bowel perforation, adhesions, and fistula. However, even if your device was not recalled, you may still be eligible for a lawsuit.
Major Hernia Mesh Recalls:
Recalled device(s): Bard Composix Kugel Mesh X-Large Patch Oval With EPTFE
Manufacturer: Davol, Inc., Sub. C. R. Bard, Inc.
Date Initiated: December 22, 2005
Class: Recall Class 1
Reason: Potential for memory recoil ring to break, leading to bowel perforation or chronic enteric fistula
Recall Status: Completed
Recalled device(s): C-QUR Mesh, C-QUR TacShield Mesh, C-QUR V-Patch Mesh, C-QUR Edge Mesh
Manufacturer: Atrium Medical Corporation
Date Initiated: July 19, 2013
Class: Recall Class 2
Reason: Additional instructions are required due to the tendency of coated mesh to adhere to inner packaging when exposed to high humidity.
Recall Status: Terminated on March 18, 2016
Recalled device(s): Proceed Surgical Mesh, Product Codes PCDB1, PCDT1, And PCDJ1. Ethicon Inc., Johnson & Johnson ...
Manufacturer: Ethicon, Inc. (Johnson & Johnson)
Date Initiated: February 19, 2014
Class: Recall Class 2
Reason: Sterility concerns
Recall Status: Open/Classified
Recalled device(s): TIGR Matrix Surgical Mesh, Model No. NSTM1015, NSTM1520, NSTM2030
Manufacturer: Novus Scientific Ab
Date Initiated: July 19, 2016
Class: Recall Class 2
Reason: Additional safety warnings issued, noting the product is not suitable for repair of direct inguinal hernias and is fully resorbable. The device should also not be used where permanent support from mesh is required.
Recall Status: Terminated on December 23, 2016
Recalled device(s): PROCEED Surgical Mesh Hernia Mesh Oval, 15 Cm X 20 Cm
Manufacturer: Ethicon, Inc.
Date Initiated: October 23, 2020
Class: Recall Class 2
Reason: Hair found inside the primary packaging of a sterile device.
Recall Status: Terminated on December 21, 2022
In addition to these recalls, Ethicon’s Physiomesh Flexible Composite Mesh was removed from the market after discovering higher than normal recurrence/operation rates associated with the device.
Current/Past Hernia Mesh Information Labels
Hernia mesh manufacturers have come under scrutiny for failing to provide adequate safety information regarding their devices. Throughout the past two decades, multiple manufacturers, including C.R. Bard and Atrium Medical Corp. have had to issue recalls to update labeling and safety concerns for their products.
FDA/EPA Warnings About Hernia Mesh
The U.S. Food and Drug Administration has issued several warnings regarding the use of hernia mesh, including a risk analysis of the use of the product. In their July 2023 informational statement, the regulatory agency detailed the most common adverse event reports. Individuals indicated high rates of infection, pain, hernia recurrence, adhesion, fistula, and organ perforation. Additionally, some hernia mesh products were subject to migration and shrinkage, often requiring additional surgeries to repair or replace. The FDA noted that they had received over 55,000 adverse event reports.
What Damages Can People Sue Hernia Mesh Manufacturers For?
Individuals who have suffered serious injuries after hernia mesh implantation may be eligible to receive compensation for their damages. Damages in hernia mesh lawsuits may include financial recovery related to both economic and non-economic losses.
Damages in a Hernia Mesh lawsuit:
- Medical Bills: Hernia mesh patients may be eligible for compensation related to past and future medical expenses if the device failed or caused significant injury. Medical bills may include hospitalizations, emergency room visits, specialists, rehabilitation, prescription drug costs, therapy, and any costs related to injury treatment now or in the future.
- Loss of Income: Plaintiffs may also receive compensation related to lost income suffered as a result of the injury and the loss of future earning capacity if the condition resulted in long-term or permanent disability.
- Loss of Quality of Life, Consortium, and Support: In addition to out-of-pocket losses, hernia mesh plaintiffs may be eligible to receive financial recovery for less tangible losses, such as if the injury resulted in a diminished quality of life. If the injury resulted in a disability or disfigurement, there may be an increase in compensation due to the severity of the condition.
- Pain and Suffering: Compensation related to physical pain and emotional distress may be sought by plaintiffs who suffered mental or physical harm as a result of the mesh-related injury.
- Legal Fees and Costs: Many plaintiffs in hernia mesh lawsuits will be eligible for compensation related to their legal fees and costs in addition to any out-of-pocket expenses.
In limited cases, plaintiffs may be able to seek punitive damages. Punitive damages are designed to punish the defendant for reckless or grossly negligent actions that resulted in serious harm to consumers.

Who Qualifies to File a Hernia Mesh Lawsuit?
In order to file a hernia mesh lawsuit, each person has to meet certain eligibility requirements. You may be able to file a lawsuit if you experienced the following issues and complications resulting from those issues:
- Infection
- Mesh migration
- Adherence to other organs
- Mesh malfunction
- Mesh deterioration
- Recurrence of hernia
Individuals must have proof that they suffered a serious complication related to the implantation of a qualifying device. If you used meshes from Atrium Medical Corporation (related to their C-Qur hernia mesh), C.R. Bard (for their PerFix, Kugel, 3DMax, and Ventralex mesh), Covidien (for Parietex mesh), and Johnson & Johnson (for Ethicon Physiomesh), you may be eligible to file a lawsuit.
What Proof Do You Need to File a Hernia Mesh Lawsuit?
To prove your hernia mesh lawsuit, you will need to gather evidence that helps to strengthen your claim. Evidence that may prove critical to your case includes medical records showcasing your initial hernia repair with a qualifying mesh implanted, subsequent side effects or harm related to the mesh implantation, and surgical intervention to repair or replace the device.
Evidence of Hernia Mesh Use and Timeline of Use
- Proof of Qualifying Hernia Mesh Product: Not all hernia mesh products are subject to liability in the current lawsuits. For a successful claim, you must be able to show that you had a qualifying hernia mesh device implanted into your body.
- Proof of Timeline: Pre-existing conditions may disqualify you from eligibility. Medical records must show a definitive timeline of when the mesh was implanted when injuries occurred, and the progression of the complications.
Evidence About Your Diagnosis and Treatments
- Medical History: Complete medical records often prove critical in hernia mesh lawsuits. The records should document the dates of surgery, any side effects, and the diagnosis of a mesh-related complication.
Evidence Showing Your Losses
- Employment Records: To prove losses related to income and future earning capacity, you must be able to prove what you made prior to the injury and any reduction in pay as a result of your inability to continue performing your duties. This may also help to prove a diminished quality of life-related to your injuries.
Testimony From You and Your Family
- Personal and Witness Testimony: Your account and the accounts of those closest to you can help to establish how the injury impacted your daily life. Statements from family, friends, and coworkers can assist in substantiating your claim.

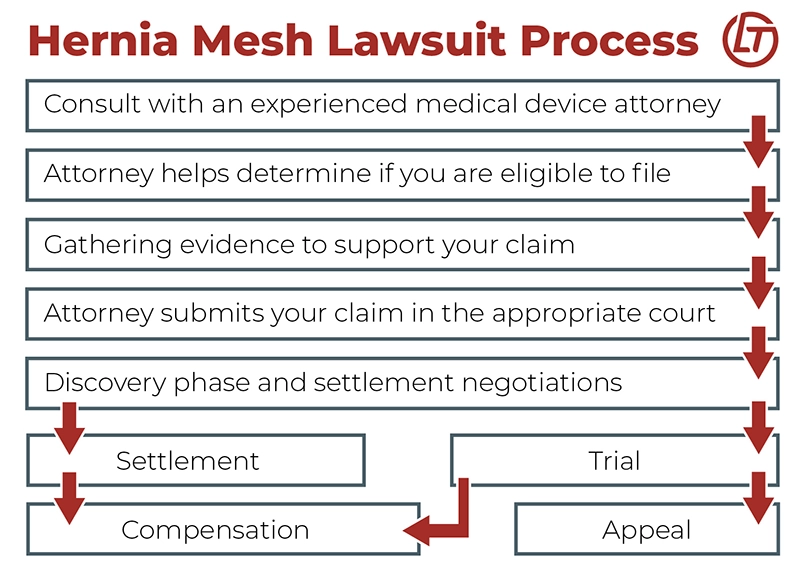
7 Steps to File a Hernia Mesh Lawsuit
There are several steps to filing a hernia mesh lawsuit. These steps include retaining a qualified attorney, establishing eligibility, gathering the necessary evidence, and filing a claim in the appropriate courthouse. It is imperative to act quickly, as some hernia mesh lawsuits are in the mid-to-late stages, meaning the time to file a claim may be running out.
These are the 7 steps for filing a hernia mesh lawsuit:
- Consult with an experienced attorney: Prior to filing a claim, it is essential to consult with an attorney who is well-versed in hernia mesh lawsuits and medical device litigation. An attorney can help guide you through the legal process and ensure all requirements are met in your case.
- Establish eligibility: During your initial case review, the attorney may review your medical history to determine whether you are eligible to file a lawsuit. Not all complications related to hernia mesh implantation will qualify.
- Collect evidence: Once eligibility is established, your attorney will aid you in collecting the evidence necessary to support your case. Critical documentation includes a complete copy of your medical records, proof of revision surgery or other treatment, and evidence of actual damages.
- File the claim: With the necessary evidence gathered, your attorney can now file the claim in the appropriate courthouse. Your legal team will review the case to ensure that it falls within the statute of limitations and that all requirements are met prior to filing. Your case may be eligible to join an existing multidistrict litigation.
- Discovery Phase and Negotiations: Once the lawsuit is filed, the case will enter the discovery phase. During this phase, both parties have the opportunity to review and exchange information. They may also enter negotiations to determine if a settlement agreement can be reached.
- Set for Trial: If a settlement cannot be negotiated, the matter may be set for trial. At trial, a judge or jury will determine the outcome of the case.
- Resolution: Depending on the outcome of the case, you may receive compensation based on the settlement agreement or jury award, or you may have the opportunity to appeal an adverse decision.
What to Expect When Filing a Hernia Mesh Lawsuit
It is important to note that hernia mesh lawsuits may take several months to a year or more to file. The process can be lengthy, but agreeing to a settlement may result in a faster resolution. The amount of compensation you may receive depends on a variety of factors, including the severity of your injury and whether your case goes to trial. Most attorneys handle hernia mesh lawsuits on a contingency fee basis, meaning you pay no upfront fees.
What is the Deadline for Filing a Hernia Mesh Lawsuit?
The deadline for filing a hernia mesh lawsuit depends on the statute of limitations that governs your case. Statutes of limitation are state-specific and can range from 1 to 6 years, although most are between 1 and 3 years. Typically, the amount of time you have to file a claim begins from the date of the injury or the date you discover the mesh implant caused your injury.
Special circumstances such as incapacitation or being a minor at the time of the injury may extend the amount of time you have to file a lawsuit. Due to the complex nature of these deadlines, it is critical to consult with an attorney as soon as an injury is discovered or diagnosed.
Hernia Mesh Settlements and Awards
Hernia Mesh settlements have ranged between $50,000 and over $1 million. There are several considerations that are taken into account to determine the value of a case, including the severity of the injury, the jurisdiction where the case is being heard, and any prior settlements.
Average Settlement Amounts
Most hernia mesh lawsuits are expected to settle between $65,000 and $80,000, although the amount may vary depending on the severity of the injuries. Some hernia mesh manufacturers have entered into confidential settlement agreements with plaintiffs; however, there have been multiple verdicts in bellwether trials against C.R. Bard. Only one of the four cases went in favor of the defense.
Jury Awards in the Bard Bellwether Trials and other cases:
- Stinson v. Davol, Inc., et al. - $500,000
- Trevino v. Bard - $4.8 million
- Milanesi v. C.R. Bard - $250,000
Factors Affecting the Value of a Hernia Mesh Lawsuit
There may be several factors that can affect the value of a hernia mesh lawsuit. These factors include the severity of the injury, impact on a person’s quality of life, and any proven misconduct by the manufacturer. The more profoundly a person is affected by their mesh-related injuries, the greater potential compensation. For instance, a person who has had multiple revision surgeries to repair or replace defective mesh is more likely to receive a higher payout compared to someone who had minor injuries such as constipation or a mild infection.
Learn More About Hernia Mesh Lawsuits From Our Legal Team
Hernia mesh failures can negatively impact nearly every aspect of a person’s life. From their inability to work to living with chronic pain, these injuries can be long-term and, in some cases, permanent. Individuals who have suffered harm from a hernia mesh implant are encouraged to consult with an experienced attorney as soon as possible to determine whether they may be eligible for compensation.
Get A FREE Case Review
Contact Us TodayThe average payout of a hernia mesh lawsuit is expected to be between $65,000 and $80,000. However, some cases have the potential to receive significantly more based on the severity of the injuries.
Hernia mesh lawsuits are still active throughout the United States. It is expected that thousands of plaintiffs may still be eligible for compensation.
The hernia mesh lawsuit is ongoing, and all four multidistrict litigations remain active. However, some litigation is in the mid-to-late stages, with global settlements expected to be announced soon.
Individuals who suffered adverse reactions to a hernia mesh implant may be eligible to file a lawsuit. However, not all injuries will qualify. It is essential to discuss your case with a knowledgeable attorney to determine your eligibility.
The deadline to file a hernia mesh lawsuit depends on a number of factors, including the date you discovered the injury, the jurisdiction where your case was filed, and whether any exceptions apply. In most cases, the statute of limitations or deadline to file a lawsuit is 1 to 3 years from the date you discovered the injury.
Several hernia mesh lawsuits are in their mid-to-late stages, although they are still active lawsuits with new claims continuing to be filed.
Thousands of cases have been consolidated into four multidistrict litigations. MDLs are designed to help streamline the pre-trial process.
It is expected that there will be thousands of hernia mesh settlements in 2025, as one of the largest manufacturers recently agreed to a global settlement.
Contact Us Today
"*" indicates required fields