Get a FREE Case Evaluation
"*" indicates required fields
Quick Facts
- What is a medical device? Any instrument, machine, implant, or testing and diagnostic equipment that is used to treat, cure, prevent, mitigate, or diagnose a disease or condition in humans
- Common types of dangerous medical devices: Hernia mesh, prosthetics, ports, and implants
- Common medical device failures: Infection, failure, breakage, shrinkage, migration, rejection, and allergic reactions
- Regulator of medical devices in the U.S.: U.S. Food and Drug Administration
- Medical device lawsuits filings: Lawsuits are typically filed against manufacturers who knew (or should have known) their devices harmed people
Defective Medical Device Lawsuits
Get the Latest News On Lawsuits About Dangerous Medical Devices
At Lawsuit Tracker, we share legal updates and information about medical devices and products that have harmed people. Medical device manufacturers and doctors have a responsibility to protect and warn consumers. When those companies and physicians fail to do so, the legal system is there for people to pursue justice.
Our team of legal professionals is here to provide relevant and up-to-date information to consumers. As lawsuits against pharmaceutical companies progress through our legal system, our team shares important information and insights.
Our site is intended to help consumers in multiple ways. If a medical device has harmed you or a loved one, we share important information and offer legal support through our legal partners. If you or a loved one is currently using a dangerous medical device, we share information about what to monitor for. That way, you can act quickly if you experience symptoms of a dangerous complication.
If you have been harmed and are considering taking legal action against the medical device manufacturer or physician who harmed you, our attorneys are ready to fight for you.
Get A Free Case Review
Contact Us TodayThis page is designed to help consumers understand how medical device manufacturers make errors and what consumers can do about it. We want to empower people with knowledge—knowledge about their current and potential injuries and knowledge about their legal rights as a consumer.
Our Active Lawsuits
At Lawsuit Tracker, we report on dangerous medical device lawsuits that matter to consumers. When people are harmed, our legal partners fight for them. If you used one of the medical devices listed below and were injured, we want to hear from you.
On this page:
Get the Latest News On Lawsuits About Dangerous Medical Devices
Why People Sue Medical Device Manufacturers
People Are Harmed by FDA-Regulated Medical Devices
How Medical Device Companies Can Be At Fault
Types of Lawsuits People File Against Medical Device Manufacturers
How Our Legal Partners Can Help People Harmed by Medical Devices
Why People Sue Medical Device Manufacturers
Medical devices go through a rigorous government-approval process. However, the process isn’t perfect and some complications may take a long time to present themselves. When a dangerous medical device or product harms people, those people may have the right to file lawsuits against the manufacturer.
There are multiple reasons a patient would file a lawsuit against a company that makes medical devices or products:
- The design of the device or product proved to be defective
- The device was designed properly, but an error occurred during the manufacturing process
- The device’s warning label did not properly warn patients of a potential injury
Some design defects are not discovered before a medical device is on the market. Some defects emerge over time, after patients have the devices in their bodies for an extended time. A device can break down in unpredictable ways, causing patients harm.
Additionally, a lot can go wrong during the medical device manufacturing process. Problems can occur during the assembly of the product, the packaging of the product, the storage of the product, and the transportation of the product
When design or manufacturing errors happen, people can be harmed by the medical devices that were supposed to help them. From devices that help doctors administer drugs to devices implanted in a patient’s body, things can and do go wrong—sometimes with serious consequences to patients.
Why People Sue Doctors After Experiencing Injuries From Medical Devices
Sometimes people are harmed by medical devices because their doctors do not follow the protocols for using or implanting a device. When this happens, patients can experience severe complications or injuries. Those patients may file lawsuits against their medical providers.
In some lawsuits, it is possible for a physician and a medical device manufacturer to be listed as defendants. In such cases, there was an error or negligence from more than one party.
These lawsuits can be complicated, so choosing an experienced attorney is important to filing a successful lawsuit. Our legal partners understand how people are harmed by medical devices and who should be held liable. Contact us to learn more about what our team can do for you.
What Is a Medical Device?
In simple terms, a medical device is an item that is used for diagnosing, curing, mitigating, treating, or preventing a disease. A medical device could be a simple product like gauze or a more complex product like a robotic machine used in a major surgery.A medical device may be used on the outside of a body, attached to the body, or implanted within the body. Medical devices can help our bodies heal, test for diseases, inject critical medications, or perform complex surgeries. Medical devices include things like insulin pumps, hernia meshes, breast implants, hearing aids, and joint-replacement components. The FDA defines medical devices under the Food, Drug, and Cosmetic Act, which is codified into Title 21 Chapter 9 of the United States Code.
“Per Section 201(h)(1) of the Food, Drug, and Cosmetic Act, a device is: An instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article, including a component part, or accessory which is:
- (A) recognized in the official National Formulary, or the United States Pharmacopoeia, or any supplement to them,
- (B) intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals, or
- (C) intended to affect the structure or any function of the body of man or other animals, and which does not achieve its primary intended purposes through chemical action within or on the body of man or other animals and which is not dependent upon being metabolized for the achievement of its primary intended purposes. The term “device” does not include software functions excluded pursuant to section 520(o).”
Medical devices are different from drugs in that they do not need to be metabolized or altered by a human or animal system to be effective. For example, an insulin pump is a medical device, while the insulin it injects is the drug. The device does not need to be digested or processed by a human to function.
Examples of Other Regulated Medical Products
In addition to devices, the FDA regulates other products that people rely on throughout their lives. Those products include:
- Biologics like blood components, plasma, and gene therapy products
- Special medical foods and infant formula
- Cosmetics, some hygiene products, hair dye, and tattoos
- Additives used in foods and beverages
However, these devices and products can and do fail, despite federal regulations. There are many reasons for these failures, which put millions of Americans in harm’s way each year.
People Are Harmed by FDA-Regulated Medical Devices
The FDA is the federal agency tasked with protecting American consumers from dangerous drugs, foods, and medical products. The FDA has many regulations for medical device manufacturers. The FDA regulates both new products and products that have been available for years.
The FDA’s Mission:
“The Food and Drug Administration is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological products, and medical devices; and by ensuring the safety of our nation’s food supply, cosmetics, and products that emit radiation.”
A lot of people think that if a medical device or product is approved by the U.S. Food & Drug Administration (FDA), then it must be safe. It is not always that simple. According to an analysis of FDA data, in the decade preceding 2020, medical devices were linked to 1.7 million injuries and 83,000 deaths in the United States. The FDA had approved and monitored each of those devices, but patient injuries still occurred.
Despite a robust approval and monitoring process, dangerous medical devices go to market and are used by consumers. Over time, device components may degrade, leading to complications. In other circumstances, chemical reactions have unintended consequences on devices. Sometimes, errors in the manufacturing process lead to device malfunction. The end result is people being harmed by defective medical devices.
The FDA’s Role in Protecting People from Dangerous Medical Devices and Products
When a medical device manufacturer wants to produce a new medical device or product, that product must go through the FDA’s Device Development Process. That process has five steps:
- Step 1: Device Discovery and Concept
- Step 2: Pre-Clinical Research – Prototype
- Step 3: Pathway to Approval
- Step 4: FDA Review
- Step 5: FDA Post-Market Safety Monitoring
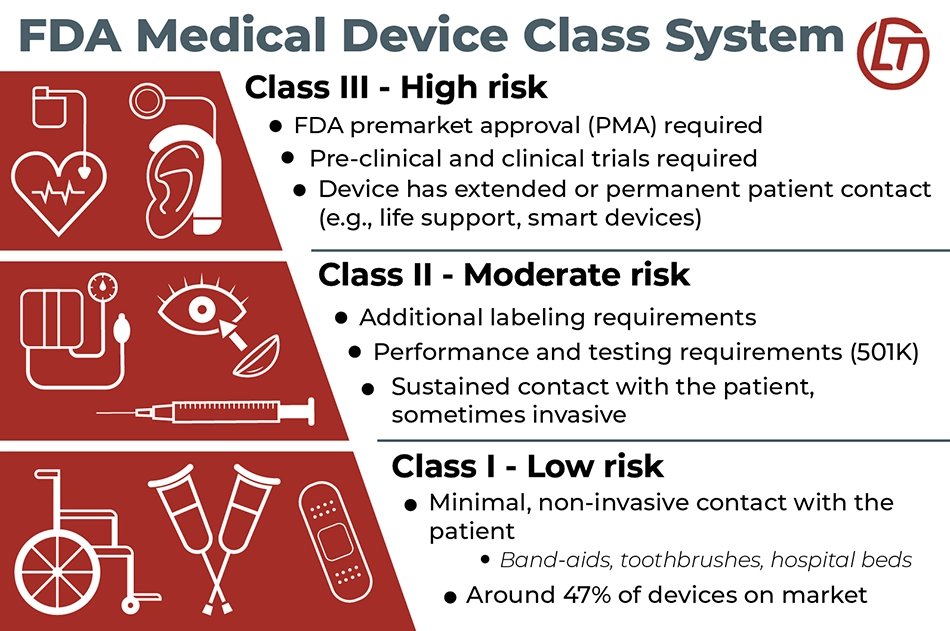
During the first step of the process, called “Device Discovery and Concept” the new device is classified as Class 1 or Class 2. Many medical devices that eventually cause injuries are given a specific classification called “Class 2: General Controls With Special Controls.” Devices in this class have additional controls, such as:
- Labeling requirements (information that must be included on a product label)
- Device-specific mandatory performance standards
- Device-specific testing requirements
Even with these safety measures in place, people still experience injuries and serious side effects from medical devices and medical products. When devices and products injure consumers, the FDA wants to know about it. To monitor these potential injuries and complications, the FDA relies on an adverse-event-reporting system and ongoing research from the scientific community.
MedWatch: The FDA Safety Information and Adverse Event Reporting Program
When patients are harmed by medical devices or therapeutic biologic products, they can report their injuries to the FDA. Patients and medical professionals can report concerns through the MedWatch reporting program. Once reported, the FDA uses a database called FAERS (Adverse Event Reporting System) to document and track each of these complaints.
The FDA reviews each of these complaints. Depending on the severity and frequency of the injury, the FDA may launch an investigation. The manufacturer also receives complaints from this database. These complaints are intended to warn device manufacturers about potential issues with their products.
Unfortunately, device manufacturers do not always act in the best interests of the public. Sometimes, they wait for the FDA to mandate a warning label update or the removal of a device from the market. This means consumers do not get proper warnings about devices that can hurt them.
If you experienced injuries after using a medical device or therapeutic biologic product, you can file a complaint in FAERS.
Medical Studies and Post-Market Surveillance
The medical and scientific research communities monitor the safety and effectiveness of medical devices and products. Scientists, doctors, medical engineers, and researchers monitor outcomes for patients who rely on medical devices as part of their treatment plans.
Any company that makes or distributes a medical device or therapeutic product must follow post-market FDA requirements. These FDA regulations are rules for how manufacturers continue to market their drugs and conduct post-market surveillance. Device manufacturers are also required to track device malfunctions, serious injuries, and deaths. They are also required to conduct continuing medical studies to monitor their products.
However, medical device manufacturers do not always put the American consumer first. Sometimes, they choose to sponsor favorable medical studies about their products. Sometimes, they take too much time to report safety concerns about their devices. This puts patients at risk of being harmed by medical devices before they know about potential dangers.
Read more about how the FDA uses scientific research in its approval and surveillance processes.

Are Drug Companies Required to Protect Consumers from Dangerous Medical Devices and Products?
The FDA is the government regulatory body in charge of protecting people from dangerous medical equipment and products. Through regulations and processes, they work to ensure the safety of devices that are on the market and devices that may one day be on the market.
The FDA places many regulations on device manufacturers who want to bring a product to the market and keep it on the market. These requirements are intended to protect as many people as possible from dangerous medical devices. The regulations for medical device companies include rules for designing, manufacturing, storing, transporting, selling, marketing, and monitoring any FDA-approved device.
Drug Company’s Rules for Marketing and Advertising
Medical device manufacturers have to adhere to strict rules when creating any marketing material that mentions their devices. Even if a device has serious side effects or risks, its manufacturer can market that device, assuming they received proper approvals. However, they have to adequately present those risks to doctors and patients. Similarly, the benefits should outweigh the risks for the majority of patients using a specific device. Said simply, just because a device is being marketed to consumers, that doesn’t mean it is safe for everyone.
Does Federal law ban ads for drugs that have serious risks?
“No. Federal law does not bar drug companies from advertising any kind of prescription drugs, even ones that can cause severe injury, addiction, or withdrawal effects.”
Source: FDA’s prescription drug advertising questions and answers
Responding to Reports of Adverse Drug Events
Mechanisms like MedWatch and the FAERS record possible injuries and complications experienced by doctors and patients while using medical devices. When numerous or very serious complaints are received, the device manufacturer is required to take action. Some of the actions include:
- Conducting scientific investigations to follow up on the concern
- Updating a device’s warning label
- Issuing a safety alert to physicians and/or the public
- Voluntarily or involuntarily removing a device from the market
Medical Device Labels and Informational Inserts
When a patient or doctor uses a medical device, that device comes with information about things like dosing, side effects, things to watch for, and important information for using or implanting the device. The device manufacturer has to include information for medical professionals and patients. Based on this information, patients and their medical teams can make informed choices about whether or not a device will benefit the patient and the potential dangers associated with the device.
However, some medical device manufacturers fight to keep the most dire warnings off a device’s warning label. When they do this, innocent people are harmed by devices that were intended to help them.
How Medical Device Companies Can Be At Fault
If a company does not meet its regulatory requirements, they can be held legally liable for the injuries they cause. When a medical device manufacturer does not take the proper steps to protect the American public, they hurt people. These people are often already dealing with serious diseases or medical issues. There are many reasons a drug company may be held legally responsible for injuring someone.
For each device a company designs or manufactures, they are required to exercise reasonable care of their medical device in the following ways:
- Design
- Formulation
- Manufacture, storage, and distribution
- Testing
- Inspection
- Packaging and labeling
- Marketing, promotion, advertising, and sales
- Research and testing of post-approval safety
If a medical device manufacturer does not uphold reasonable care in each of these areas, they may be held liable for the injuries caused by their devices.
During the litigation of a civil lawsuit, it can be determined that drug companies knew (or should have known) about the dangers their devices posed and failed to take action. Instead of properly warning physicians and patients about serious side effects and complications, they chose to keep producing and selling dangerous devices.
What Are the Reasons People Sue Medical Device Companies?
There are many reasons a patient may decide to file a lawsuit against a medical device manufacturer. When someone is seriously harmed by a medical device, financial compensation may be the only way to provide for themselves or their families during an incredibly difficult time.
But, what grounds do injured people file lawsuits on? What are the legal reasons people sue medical device manufacturers for? It all comes down to allegations of not taking the proper actions. Essentially, people sue medical device companies for things like failure to take proper actions, negligence, product defects, and not making device safety their first priority. Here are some of the legal allegations filed against manufacturers of dangerous medical devices:
- Failure to Warn
- Negligent Failure to Warn
- Design Defect
- Negligence
- Negligent Design Defect
- Negligent Misrepresentation
- Fraudulent Misrepresentation
- Breach of Express Warranty
- Breach of Implied Warranty
All of these accusations imply that a medical device manufacturer failed to do something and that failure hurt someone, often in a serious way.
Types of Lawsuits People File Against Medical Device Manufacturers
When medical devices hurt or kill people, those people or their families have several options for legal recourse. These options include filing a personal injury lawsuit, filing under a multidistrict lawsuit, or joining a class-action lawsuit. Each of those options involves civil lawsuits.
Civil Lawsuit:
A type of legal process that settles a dispute between two parties. In a civil lawsuit, an injured party (Plaintiff) seeks damages (compensation) from a defendant. A civil lawsuit does not involve criminal actions or result in jail time for the defendant.
Medical Device Manufacturers and Mass Torts
When a medical device fails and causes injuries, those injuries are often experienced by many people. When many people experience the same injury from the same device and those people file lawsuits, those lawsuits may become part of a mass tort.
Tort:
A tort is a civil wrongdoing that results in harm to a person or their property.
Mass tort:
A type of civil lawsuit where multiple plaintiffs (injured parties) seek damages from the same defendant(s). The lawsuits are consolidated into the same court, but each person’s case is presented individually and compensation varies from case to case.
Mass torts are intended to streamline the legal process for the plaintiffs, defendants, and courts. The early steps of a lawsuit (e.g., discovery) are established by bellwether trials. So people who file lawsuits after the bellwether trials conclude benefit from a streamlined legal process.
The goal of a mass tort is to combine multiple, similar court cases into a single legal proceeding. In this proceeding, each case is presented on its own merits. This means that how someone was injured and how severely they were injured would determine how much they might receive from a settlement.
Mass Tort Fact:
Successful plaintiffs’ compensation is based on their specific injuries and circumstances related to medical device failure. A plaintiff’s lawyer fights for the maximum damages their client is allowed.
Medical Device Manufacturers and Multidistrict Litigation
When medical devices harm multiple people in a similar way, many people take action by filing civil lawsuits. These lawsuits may be filed in state or federal courts. Sometimes, those cases are consolidated as part of a single multidistrict litigation (MDL). An MDL is a type of civil lawsuit that combines cases from multiple jurisdictions into a single federal court.
MDL Fact:
Unlike a class action lawsuit, successful plaintiffs in an MDL receive compensation based on the extent of their individual injuries. That money often comes from a settlement, but settlements vary from person to person.
Drug Companies and Personal Injury Lawsuits
When someone experiences a serious injury due to a medical device, they and their attorney may file a civil lawsuit against the device manufacturer. In an individual personal injury lawsuit, the injured person is not part of a class-action or MDL litigation. The person who was injured (the plaintiff) files a stand-alone case against a defendant. In this type of lawsuit, the plaintiff and their attorney have to go through the entire legal process on their own. However, these lawsuits, when successful, can result in large payouts.
Drug Companies and Class Action Lawsuits
Sometimes, actions against medical device manufacturers are brought as a single class-action lawsuit. In a class-action lawsuit, injured parties can join a class, which is represented by a single representative. That class representative files a single complaint and tries their case. If that representative is successful, everyone who is part of the class could receive the same compensation.
Class Action Fact:
Each plaintiff in a class action lawsuit typically receives the same amount of money. Their individual circumstances are not considered by the courts because their cases are tried as one.
How Our Legal Partners Can Help People Harmed by Medical Devices
If you or a family member experienced serious injuries from a medical device, our team may be able to help you recover money. At Lawsuit Tracker we are a team of legal partners who represent people harmed by dangerous medical devices and products.
Our lawyers can help you and your family regain stability. Our job is to prove to the courts how your injuries were caused by a medical device company’s negligence or failures. Our legal partners have a wealth of experience in mass torts, and we take our job of helping you very seriously.
Attorneys for People Harmed by Dangerous Pharmaceutical Drugs
If you have been harmed by a medical device and are interested in learning more about your legal options, contact our team. We can work with you to determine if you qualify to file a claim. If you decide you are ready, we are ready to fight for you and help you get the compensation you deserve.
Get A Free Case Review
Contact Us TodayWhat Compensation Can People Harmed by Dangerous Medical Devices Receive?
When a dangerous medical device harms someone, it can be a life-changing event. Injured people can experience a loss of income along with mounting medical bills. The losses can go beyond financial, as changes in mental health and relationships can follow. Our team is here to help you offset those losses. If you choose to file a case against a medical device manufacturer, you may receive compensation related to the following damages:
- Medical expenses (e.g., hospital stays, treatments, doctor visits, testing, rehabilitation)
- Lost wages
- Pain and suffering
- Emotional distress
- Future medical costs
- Loss of consortium (negative impacts to your relationship with your spouse)
Our team understands the serious impacts these losses can have on your life. We are ready to fight for you.
Related Content
Contact Us Today
"*" indicates required fields