Get a FREE Case Evaluation
"*" indicates required fields
Quick Facts: Spinal Cord Stimulator
- Brand Names: Eterna, WaveWriter, Intellis, Vantis, Proclaim, Infinity, Precision, Restore, Inceptiv, Itrel, Synergy, HF10 (Senza), Evoke
- Device Used For: Chronic back and leg pain
- Product type: Medical device
- Manufacturers With Lawsuits: Abbott Laboratories (St. Jude), Boston Scientific, Medtronic, FDA, Saluda, and Nevro
- Potential Severe Complications: Paralysis, numbness, loss of feeling, lead migration, nerve damage, new or worsening pain, and electrocution
- Lawsuit Type: Individual lawsuits with the potential to be consolidated
- Lawsuit Status: Early stages
Spinal Cord Stimulator Lawsuit – April 2026 Update
People are filing spinal cord stimulator lawsuits, alleging that these devices caused injuries instead of relieving chronic pain. Spinal cord stimulator lawsuits have been filed against companies like Medtronic, Boston Scientific, Abbott, and Nevro. In their lawsuits, spinal cord stimulator (SCS) patients allege that due to device defects, they were electrocuted, experienced paralysis, experienced new or worsening pain, or suffered other complications.
Most of these lawsuits are in their early stages, and a motion has been filed to consolidate them into MDL#3181 (In re: Abbott and Boston Scientific Spinal Cord Stimulator Products Liability Litigation) in the Northern District of Illinois. However, the Joint Panel on Multidistrict Litigation (JPML) has not yet accepted this request.
As of April 2026, there are about 15 lawsuits listed in the centralization request for spinal cord stimulator lawsuits. However, people have filed and continue to file personal injury lawsuits due to alleged injuries caused by their spinal cord stimulators. An attorney can advise those affected on their legal options.
Spinal Cord Stimulator Lawsuit Timeline & News – April 2026
March 5, 2026: Women Sue Abbott Laboratories Over Allegedly Defective Spinal Cord Stimulators
Three women have filed lawsuits against Abbott Laboratories, accusing the company of selling defective spinal cord stimulators. All three women had Abbott’s spinal cord stimulator implanted, and they all say they were told the device would relieve their pain. Collectively, the women have suffered from painful, embarrassing, or life-altering complications they believe are related to the spinal cord stimulator. Some of their symptoms include urinary incontinence, new or worse nerve pain, electric shock sensations, and “numbness or loss of feeling” in their extremities. The women say Abbott failed to warn them of the likelihood of these and other complications from the spinal cord stimulator.
February 20, 2026: Spinal Cord Stimulator Patients Ask Panel to Consolidate Their Claims Into MDL
The plaintiffs in about 15 lawsuits have asked the United States Judicial Panel on Multidistrict Litigation (JPML) to create a consolidated MDL for spinal cord stimulator claims against Boston Scientific, Abbott, and the FDA. The plaintiffs all received a spinal cord stimulator implant manufactured by one of these two companies, and they suffered from unexpected and serious side effects. They believe they should have been told about the risks of and the high number of adverse events that spinal cord stimulator patients have reported. The plaintiffs are also suing the FDA, claiming that it should have taken more steps to make sure that Abbott and Boston Scientific’s spinal cord stimulators were safe even after the companies modified the original designs.
February 23, 2026: Medtronic and FDA Face Legal Action Brought by Spinal Cord Stimulator Patient
A spinal cord stimulator patient is suing Medtronic and the FDA (Case No. 3:26-cv-00205). According to the man’s legal complaint, Medtronic did not properly warn him about complications and low success rates that patients may experience if they get a spinal cord stimulator. The man also claims that Medtronic’s representatives practiced medicine without authorization. The man’s lawsuit also accuses the FDA of approving over 400 pre-market modifications to Medtronic’s spinal cord stimulator design without requiring the company to submit a new application or conduct additional clinical trials. These pre-market modifications were submitted after the FDA approved Medtronic’s original spinal cord stimulator design in 1986.
January 27, 2026: Another Patient Sues Boston Scientific and the FDA After Spinal Cord Stimulator Complications
A man received a spinal cord stimulator made by Boston Scientific and was led to believe that it would significantly and reliably improve his chronic pain levels. The man’s legal complaint alleges that the Boston Scientific representative made the man believe that the procedure was safe. According to legal filings, after receiving the spinal cord stimulator implant, the man experienced electrical shocking sensations, battery warming, nerve pain, and “left lower extremity pain.” The legal complaint claims the man had his spinal cord stimulator removed, and he is still experiencing pain issues. In his lawsuit, the man alleges that Boston Scientific’s representatives did not warn him about possible complications associated with spinal cord stimulators, including the ones he experienced.
October 17, 2025: WaveWriter Patient Sues Boston Scientific, FDA for Not Warning of Spinal Cord Stimulator Risks
A woman who received a WaveWriter spinal cord stimulator manufactured by Boston Scientific has filed a lawsuit against it and the organization that approved the device’s design. According to her lawsuit, the woman had a spinal cord stimulator implanted to help relieve chronic pain. Before she had the device implanted, she participated in a test trial, in which an external device improved her pain. However, her legal complaint says that, after she had the permanent device installed, her pain got worse, and she experienced electrical shocks, the battery flipped over, and the electrical leads migrated. According to legal filings, these complications prompted her to have multiple surgeries to fix the spinal cord stimulator, which she eventually had removed. She claims that Boston Scientific’s representatives did not tell her that these and other serious complications were possible if someone received a spinal cord stimulator implant.
On this page:
Spinal Cord Stimulator Lawsuit Timeline & News – April 2026
What is the Spinal Cord Stimulator Lawsuit About?
Why Are People Filing Spinal Cord Stimulator Lawsuits?
Which Companies Are Named in Spinal Cord Stimulator Lawsuits?
What Stage is the Spinal Cord Stimulator Lawsuit In?
What Is a Spinal Cord Stimulator and How Does It Work?
Spinal Cord Stimulator Complications
Possible Injuries Caused By Spinal Cord Stimulator
Studies Linking Spinal Cord Stimulator and Serious Injuries
Spinal Cord Stimulator Recalls
FDA Warnings About Spinal Cord Stimulators
What Damages Can People Sue Spinal Cord Stimulator Manufacturers For?
Who Qualifies to File a Spinal Cord Stimulator Lawsuit?
5 Steps to File a Spinal Cord Stimulator Lawsuit
What is the Deadline for Filing a Spinal Cord Stimulator Lawsuit?
Spinal Cord Stimulator Settlements and Awards
Learn More About Spinal Cord Stimulator Lawsuits From Our Legal Team
What is the Spinal Cord Stimulator Lawsuit About?
Spinal cord stimulator lawsuits accuse device manufacturers of misleading patients about the possible results and risks associated with spinal cord stimulator (SCS) implants. Some of the patients who have filed lawsuits claim they were given an external trial version of the implant first, which helped their symptoms, but the internal, permanent spinal cord stimulator injured them.
Some of the lawsuits also describe instances where spinal cord stimulator representatives may have given patients medical advice and performed medical services without authorization. The FDA is also accused of not doing enough to make sure the modified spinal cord stimulators were safe and aligned with the originally approved design.
| Spinal Cord Stimulator (SCS) Lawsuit Information | |
| Lawsuit Name: | Spinal Cord Stimulator Lawsuit |
| Main Injuries: | New or worsening pain, electrical shock, paralysis, nerve damage, nerve pain, burns caused by battery overheating, |
| Defendants: | Boston Scientific, Nevro, Abbott, St. Jude, Medtronic, and Saluda |
| Mass tort or class action? | Motion for centralization with JPML, possible future MDL |
| Court Name (Venue): | Federal and state courts |
| Have There Been Settlements? | Yes |
| Active Lawsuit? | Yes |
Why Are People Filing Spinal Cord Stimulator Lawsuits?
People are filing spinal cord stimulator lawsuits because they say they suffered severe, unexpected, and sometimes permanent complications after receiving a stimulator implant. Some studies and public reports have suggested that spinal cord stimulators have a high rate of complications and device failure.
Some spinal cord stimulator patients say they were led to believe that these devices are safe and that the risk of complications is low. They also say they were not told about the high number of adverse events reported to the FDA about spinal cord stimulators.
Some people who have received a spinal cord stimulator implant say they experienced nerve damage, paralysis, numbness, tingling, electrocution, burns, lead migration, new or worsening pain, or other complications. Many of the people who have filed lawsuits claim the spinal cord stimulator complications have led to costly medical treatments, a loss of income, loss of quality of life, pain and suffering, and other setbacks.
Spinal Cord Stimulator Lawsuit Spotlight
Spinal Cord Stimulator Patient in Texas Sues Abbott
A person in Texas is suing Abbott Laboratories, which manufactured the Eterna spinal cord stimulator. The patient received the Eterna spinal cord stimulator on October 6, 2023, and had it removed on December 13, 2023, because the device allegedly failed.
According to their legal complaint, Abbott representatives worked extensively with the patient during the trial period and after the implant was finished. The patient also says the representatives reprogrammed the spinal cord stimulator several times, even though they do not have a medical degree.
Allegedly, the patient experienced severe pain and other symptoms due to lead migration and other complications as a result of the allegedly defective spinal cord stimulator. The patient claims that they were not warned about the risk of complications associated with spinal cord stimulators.
Which Companies Are Named in Spinal Cord Stimulator Lawsuits?
People are filing lawsuits against the companies that made or approved the allegedly defective spinal cord stimulators. These are the defendants in the Spinal Cord Stimulator lawsuit:
- Boston Scientific
- Abbott Laboratories (St. Jude)
- Medtronic
- Nevro Corp.
- Saluda Medical
- U.S. Food and Drug Administration (FDA)
Information for some of the defendants is listed below.
Defendant 1: Boston Scientific
Boston Scientific is a medical device company that was founded in 1979 in Massachusetts. In 2004, the company requested FDA approval for the first rechargeable spinal cord stimulator system, Precision, which included the Precision Spectra (released in 2013) and the Precision Montage Spinal Cord Stimulator Systems. Boston Medical developed the WaveWriter spinal cord stimulator series, which includes the Spectra WaveWriter, WaveWriter Alpha, and WaveWriter Alpha Prime systems. On its website, Boston Medical claims that its WaveWriter Alpha System is an “SCS therapy that succeeds where others fail and gets it right the first time.”
Defendant 2: Medtronic
Medtronic was founded in 1949 in Minneapolis and developed the first battery-operated pacemaker. Medtronic began manufacturing spinal cord stimulators in the 1980s. According to Blue Cross Blue Shield records, Medtronic’s spinal cord stimulator systems include Itrel, Restore, Synergy, Intellis, and Vanta. That same document indicates that Medtronic’s Inceptiv Spinal Cord Stimulation System replaced the Intellis series in April 2024. Medtronic also developed the Pisces, Specify, and Vectra spinal cord stimulator series. Medtronic received FDA approval for its first spinal cord stimulator in 1984, but it developed more spinal cord stimulator devices without requesting separate new device approvals or submitting additional clinical trial data. A number of Medtronic’s spinal cord stimulator devices and accessories have been recalled.
Defendant 3: Abbott Laboratories
Abbott Laboratories was established in 1888 and acquired St. Jude Medical in 2017. St. Jude developed several spinal cord stimulator series, which are now owned and manufactured by Abbott.For example, Abbott (St. Jude) received FDA approval for its Genesis and Eon spinal cord stimulator systems in 2001. Abbott (St. Jude) produces the Prodigy, Proclaim XR, Proclaim Plus, and Externa Spinal Cord Stimulation (SCS) Systems. Abbott’s Infinity and Proclaim series of spinal cord stimulators were recalled because patients were having difficulties getting their devices to exit MRI mode. Abbott has issued other advisories and recalls related to spinal cord stimulators and accessories that it manufactures.
Defendant 4: The FDA
The United States Food and Drug Administration is a federal agency that reviews applications for medical devices. The FDA also authorizes companies to market and sell devices and requires them to provide updated safety data after devices enter the market.
The FDA has been criticized by some people for allegedly allowing spinal cord stimulator companies to repeatedly modify their original designs, essentially turning the stimulators into different devices. Some of the legal complaints allege that the FDA should have required the companies to conduct updated clinical trials for these materially different devices.
Allegations Against Spinal Cord Stimulator Manufacturers
Some of the legal complaints filed against SCS manufacturers accuse them of the following:
- Failing to warn spinal cord stimulator patients about some of the risks of severe complications associated with the devices
- Negligently marketing the devices as safe and effective
- Allowing company representatives to practice medicine (e.g., give spinal cord stimulator patients medical advice) without authorization to do so
- Designing an allegedly defective spinal cord stimulator project
- Making numerous modifications to the original spinal cord stimulator instead of submitting a new device application
Some lawsuits also accuse the FDA of failing to properly regulate spinal cord stimulator companies. For example, some claim the FDA should not have let companies submit hundreds of modifications to their original devices.

What Stage is the Spinal Cord Stimulator Lawsuit In?
The spinal cord stimulator lawsuit is in its early stages. Public reports have begun to surface about life-altering and severe complications that some spinal cord stimulator patients have experienced. Some spinal cord stimulator patients in the United States and in other countries have begun to file legal complaints against the manufacturers of those devices.
A motion has been filed to consolidate individual lawsuits into a coordinated federal group claim (called an MDL). Spinal cord stimulator patients who believe they experienced a serious complication that they were not warned about can reach out to an attorney to begin the filing process.
What Is a Spinal Cord Stimulator and How Does It Work?
A spinal cord stimulator is a medical device that delivers weak, controlled electrical impulses directly into someone’s nerves to mask chronic pain signals. The device is implanted into someone’s lower back and spine. It contains a battery pack, electrical leads, and other components. The device is synced to an app or software that the patient or the clinician has access to.
Spinal cord stimulators were first approved by the FDA in the early 1960s as an external device. By the 1980s, permanent, surgically implanted spinal cord stimulators were available. SCS devices were designed to address chronic, intractable (unmanageable) pain disorders that involve someone’s arms, legs, or lower back. Spinal cord stimulation devices are based on the Gate Control Theory of pain, which claims you can reduce the feelings of pain by blocking the painful nerve signals with other, non-painful signals. A spinal cord stimulator is designed to send neutral or weak signals, so the brain receives those signals instead of the pain signals.

Spinal Cord Stimulator Complications
According to Public Citizen, between 2004 and 2019, the FDA received over 220,000 adverse event reports for patients who received an implanted spinal cord stimulator device. According to the same report, more than 500 of these adverse event reports involved the spinal cord stimulator patient dying or becoming paralyzed.
Less Common Complications of Spinal Cord Stimulator
- Paralysis
- Numbness
- Nerve damage
- Electrocution
- Spinal fluid leaking
Common Complications of Spinal Cord Stimulator
- Pain or discomfort after surgery
- Programming issues
- Infections
- Lead migration
- Battery overheating
- Device breaking (e.g., leads sticking out of the patient’s back)
A study published in the Medical Journal of Australia looked at spinal cord stimulator patient outcome data supplied by five health insurance companies in Australia. The researchers found that about 25% of spinal cord stimulator patients in that database required invasive surgeries to correct or remove the device. The researchers noted that most of those complications occurred within three years after the patients received the spinal cord stimulator.
Possible Injuries Caused By Spinal Cord Stimulator
Many spinal cord stimulator patients have said that they experienced severe injuries after they had the permanent device implanted. These injuries include:
- Paralysis
- Numbness
- Nerve damage
- New or worsening pain
- Battery overheating and burning them
- Electrocution
- Lead migration
Many of the spinal cord stimulator patients who have filed legal claims have said they were not told that these complications may occur.
Paralysis, Numbness, and Nerve Damage
Some spinal cord stimulator patients allege that they became paralyzed or experienced nerve damage or numbness after having the device implanted. For example, one patient became partially paraplegic after the surgery, and researchers believe it may be because the paddle electrodes on the spinal cord stimulator migrated. Some spinal cord stimulator patients have also reported ongoing and new nerve pain that does not go away with treatment
New or Worsening Pain
Spinal cord stimulator patients have alleged that they experienced new or worsening pain after having the device implanted. Chronic back pain is one of the conditions for which spinal cord stimulators have been marketed to treat. However, some patients claim that their pain levels increased and they developed new levels or sources of pain after having the spinal cord stimulator implanted. Some of the patients who have filed legal claims have alleged that this pain is ongoing and has not gone away after treatment.
Battery Overheating and Burning Patients
Some spinal cord stimulator patients have also claimed that the device’s battery overheated and burned or shocked them. For example, one man said he developed new, worsening pain after the spinal cord stimulator overheated and shocked him. He says his pain levels became so severe and debilitating that he had to file for disability, even after the device was removed. Other patients have also claimed that the spinal cord stimulator overheated or electrocuted them.
Lead Migration
Lead migration is another commonly reported complication, which has sometimes resulted in damage to the patient’s spinal cord or nerves. Some patients have reported that the leads shifted and pushed through their skin.
The FDA wants to receive reports of injuries and complications people experience after using a medical device. If you have a medical device-related injury you want to report, you or your physician can make a report in MAUDE (i.e., the Manufacturer and User Facility Device Experience (MAUDE) Database).
Signs and Symptoms of Spinal Cord Stimulator Complications
Some of the signs and symptoms of spinal cord stimulator complications include the following:
- Pain at the implantation site
- Feeling the leads through the skin
- Swelling or discoloration of skin
- Fever
- Inability to move or feel parts of the body
- New or worsening pain
- Shocking sensations
- Burning, itching, or heat coming from the device
Patients who receive a spinal cord stimulator implant and are concerned about symptoms they are experiencing should talk to their doctor right away. Patients know their bodies best. If something seems off to the patient, it is a good idea for them to tell their medical team as soon as possible.
Studies Linking Spinal Cord Stimulator and Serious Injuries
Several studies have suggested an association between spinal cord stimulators and severe injuries. For example, an observational study found that many spinal cord stimulator patients in the study had to have the device removed because of complications. Another study found that there was no significant difference between the pain levels of participants who received a working spinal cord stimulator versus a placebo device.
Important Study: Outcomes for SCS Patients in Australia
In a study called “Important Study: Spinal cord stimulation patterns of care, re-interventions, and costs for private health insurers, Australia, 2011-22: a retrospective observational study,” researchers examined an insurance dataset of about 5,839 people (4,361 of whom had a spinal cord stimulator device implanted). The goal of the study was to measure the outcomes and costs for people who received this implant. According to the results, 23.2% (nearly a quarter) of spinal cord stimulator patients in the study underwent surgery to modify or remove the spinal cord stimulator within 3 years after implantation.
| Overview: Spinal cord stimulation patterns of care, re-interventions, and costs for private health insurers, Australia, 2011-22: a retrospective observational study | |
| Description of Study: | The study examined the outcomes and costs of care for spinal cord stimulator patients using data from insurance databases. |
| Published In: | Medical Journal of Australia |
| Study Authors | Caitlin Mp Jones, Christopher G Maher, Rachelle Buchbinder, Ian A Harris, Chung-Wei Christine Lin, Christopher Hayes, Alexandra Gorelik |
| # of Participants | 5,839 |
| Findings | The authors discovered that about one in four spinal cord stimulator patients in the study needed to have surgery to correct or remove the device within 3 years after it was implanted. |
Important Study: SCS Devices vs. Placebos
In a study called “Effect of Spinal Cord Burst Stimulation vs Placebo Stimulation on Disability in Patients With Chronic Radicular Pain After Lumbar Spine Surgery,” researchers compared patient pain levels after a trial of a spinal cord stimulator or a placebo device. There were 50 patients in the trial, and each of them had a spinal cord stimulator device implanted.
Each patient went through a randomized period where the device delivered burst stimulation and another period where the device was not working (the placebo period). The researchers compared patient-reported pain levels during and after the trial period. According to the results, the spinal cord stimulator device did not perform any better than a placebo device at improving patients’ back pain levels.
| Overview: Effect of Spinal Cord Burst Stimulation vs Placebo Stimulation on Disability in Patients With Chronic Radicular Pain After Lumbar Spine Surgery | |
| Description of Study: | The study looked at whether an operational spinal cord stimulator performed better than a placebo device at improving patient pain levels. |
| Published In: | JAMA |
| Study Authors | Sozaburo Hara; Hege Andresen, RN; Ole Solheim, MD, PhD; et al |
| # of Participants | 50 |
| Findings | The authors found that an operational spinal cord stimulator did not perform better than a placebo device at improving patients’ self-reported pain levels. |
Spinal Cord Stimulator Recalls
Several spinal cord stimulators and accessories have been recalled. A number of these recalls were voluntarily initiated by the manufacturer. Here are some of the spinal cord stimulator recalls that have been reported:
- WAVEWRITER ALPHA 16 IPG KIT, WAVEWRITER ALPHA 32 IPG KIT (Class II recall)
- The Precision Spinal Cord Stimulator System (Class II recall)
- Artisan ™ (Class II recall)
- LinearTM 34 (Class II recall)
- Bionic NAVIGATOR Clinician Programmer Model Number SC71504 (Class II recall)
- Proclaim and Infinity (Class I recalls)
- Liberta (Class II recall)
Some of these recalled devices may have resulted in an increased risk of patient injuries, including the device overheating or electrocuting the patient.
FDA Warnings About Spinal Cord Stimulators
The FDA has issued several warning letters about spinal cord stimulators, including device models that have been recalled. For example, on September 13, 2023, the FDA announced a Class I recall of Abbott’s Proclaim and Infinity spinal cord stimulators because some models were getting stuck in MRI mode and could not be turned back on. The FDA has also received over 200,000 adverse event reports about patients who have had negative experiences involving spinal cord stimulators.
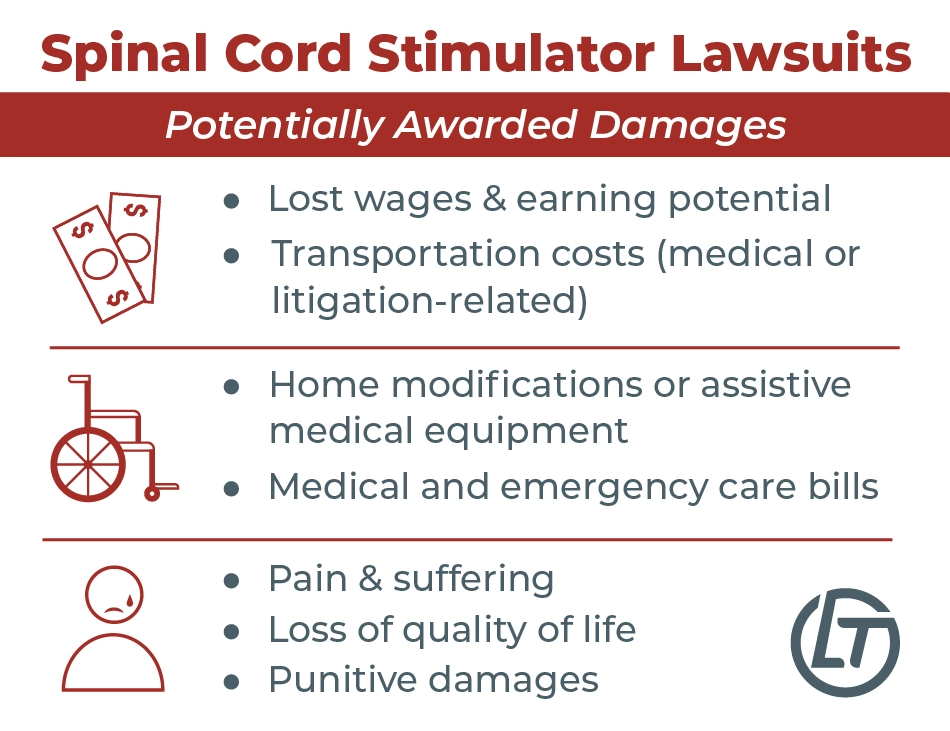
What Damages Can People Sue Spinal Cord Stimulator Manufacturers For?
Some patients may be able to receive the following compensation if they win their lawsuits against spinal cord stimulator companies:
- Past and future medical expenses
- Loss of earnings
- Loss of earning potential
- Loss of quality of life
- Pain and suffering
- Transportation costs (e.g., to and from medical appointments)
- Home modifications or assistive medical equipment costs
A qualified attorney can help affected patients estimate what their compensation package might be if they win their spinal cord stimulator claims.
Who Qualifies to File a Spinal Cord Stimulator Lawsuit?
In order to file a spinal cord stimulator lawsuit, each person has to meet certain eligibility requirements. Our legal partners are focusing on the specific injuries listed below:
- The person had a spinal cord stimulator implanted.
- The person experienced severe complications (e.g., electrocutions, nerve damage, paralysis, or burns) after the device was implanted.
The patient may still qualify to file a lawsuit even if they experienced a device-related injury that is not on the list above.
What Proof Do You Need to File a Spinal Cord Stimulator Lawsuit?
The patient’s spinal cord stimulator lawsuit needs to be supported by evidence showing that they experienced severe, unexpected complications after having the device implanted.
Evidence that can be used to support an SCS lawsuit includes:
- Medical records showing that they were implanted with a spinal cord stimulator
- Medical notes about why the device was implanted and what symptoms the patient experienced afterward
- Device and model numbers and other details about the spinal cord stimulator (including informational pamphlets given to the patient)
- Receipts, insurance claims, and after-visit summaries
- Pain diaries showing the patient’s symptoms before and after having the spinal cord stimulator implanted
- Expert testimony from medical providers and others
The patient can work with a legal team to locate, request, and appropriately use evidence to strengthen their spinal cord stimulator claim.
5 Steps to File a Spinal Cord Stimulator Lawsuit
These are the 5 steps for filing a spinal cord stimulator lawsuit:
- Schedule a free consultation with an experienced spinal cord stimulator attorney.
- Help your attorney gather and request documents needed to support your spinal cord stimulator lawsuit.
- Your attorney writes and files the legal complaint against the spinal cord stimulator company or companies.
- Your attorney attempts to settle the lawsuit with the spinal cord stimulator company.
- Your attorney takes the spinal cord stimulator case to trial if necessary to maximize your chances of winning a fair payout.
Working with an experienced legal team can help you confidently and efficiently navigate your spinal cord stimulator lawsuit.
What to Expect When Filing a Spinal Cord Stimulator Lawsuit
When filing a spinal cord stimulator lawsuit, keep in mind that you may not receive a settlement right away. It takes time to gather evidence, build a spinal cord stimulator lawsuit, and negotiate a fair payout.
Some spinal cord stimulator claims may take up to a year or more to resolve because of the administrative steps involved. Even so, these steps may be necessary to enable your legal team to develop and carry out a strong legal strategy to help tell your story and fight for a fair outcome.
What is the Deadline for Filing a Spinal Cord Stimulator Lawsuit?
The deadline to file a spinal cord stimulator lawsuit depends on the type of claim being filed and where it is filed. States have their own rules (called statutes of limitations) about when lawsuits need to be filed. These deadlines may be different for each type of claim, such as personal injury, wrongful death, product liability, or medical negligence.
There are also exceptions to these statutes of limitations, such as if the patient is incapacitated or if information is concealed from them. An attorney can help you understand what the deadline is to file your spinal cord stimulator lawsuit and how to meet it.
Spinal Cord Stimulator Settlements and Awards
The spinal cord stimulator lawsuits are in their early stages, but some individual lawsuits have resulted in settlements and verdicts. For example, some patients received settlements of more than $1 million for their spinal cord stimulator lawsuits filed against their allegedly negligent medical providers. Another man received a $1.25 million settlement after he became partially paralyzed and incontinent because of an allegedly defective spinal cord stimulator. Spinal cord settlements reflect the patient’s circumstances, the applicable law, and other individual factors. Past results do not guarantee future outcomes.
Average Settlement Amounts
Some spinal cord stimulator settlements may reach up to $250,000 or more if the patient wins their claim and has suffered significant losses. However, this is a broad estimate for illustrative purposes only. Settlements and verdicts are highly individualized and are not guaranteed.
Factors Affecting the Value of a Spinal Cord Stimulator Lawsuit
Some factors that may affect the value of someone’s spinal cord stimulator lawsuit include the following:
- The severity of their injuries
- Whether they file their claim before the statute of limitations expires
- The strength of their case
An attorney can help people estimate and fight for fair compensation for their individual spinal cord stimulator claim.
Learn More About Spinal Cord Stimulator Lawsuits From Our Legal Team
If you experienced complications that you believe were caused by your spinal cord stimulator, get in touch with our legal partners today. You may be entitled to compensation for your losses, including your medical bills, lost wages, and pain and suffering.
Our legal partners have over a decade of experience fighting for people who were harmed by defective medical devices and dangerous drugs. They can help you understand if you have a spinal cord stimulator claim and, if so, how you can protect your legal rights.
Get A FREE Case Review
Contact Us TodayA spinal cord stimulator lawsuit is filed because someone experienced an injury alleged to have been caused by an SCS device. These lawsuits accuse companies of failing to warn people about the risks associated with spinal cord stimulators.
Some people are filing lawsuits because they experienced severe and unexpected complications they believe were caused by their spinal cord stimulator implant.
Patients should talk to their doctors about whether a spinal cord stimulator is safe in their situation. The FDA has received over 200,000 adverse event reports about spinal cord stimulators.
Compensation that may be available to patients who received a spinal cord stimulator implant includes lost wages, medical expenses, and pain and suffering.
Boston Scientific, Abbott (St. Jude), Medtronic, and Saluda are some of the companies being sued in spinal cord stimulator lawsuits.
It depends. Many spinal cord stimulator lawyers take these cases on a contingency basis, so the client does not have to pay any upfront legal fees.
It depends. Some spinal cord stimulator lawsuits take up to a year or more to resolve.
Individual and group claims have been filed about spinal cord stimulators. A request was filed to consolidate many spinal cord stimulators into a consolidated multidistrict litigation. However, the request has not been granted yet.
The FDA has received over 200,000 reports of adverse events related to spinal cord stimulators.
Contact Us Today
"*" indicates required fields